QUESTION IMAGE
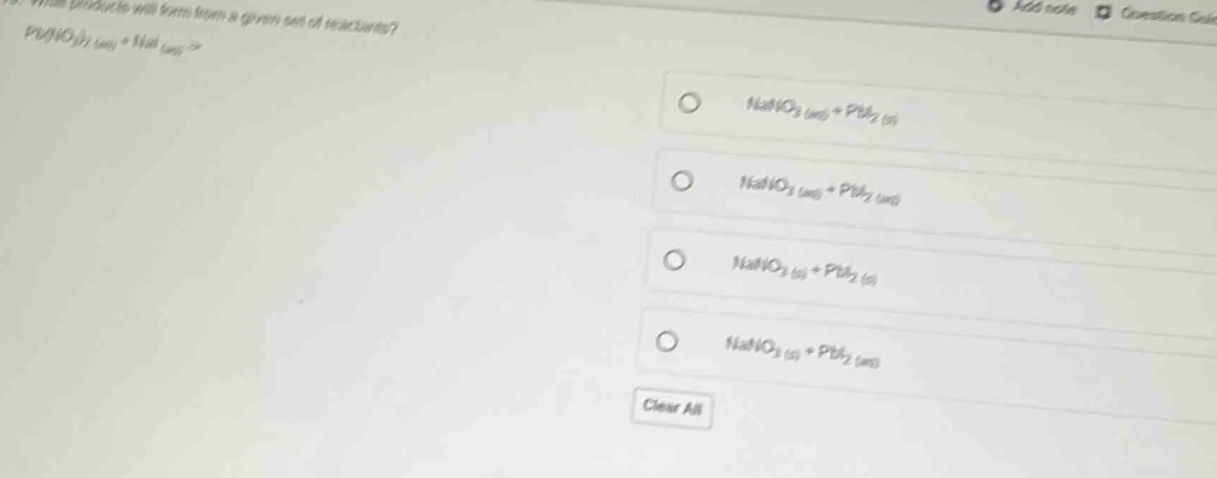
Question
what products will form from a given set of reactants?
pb(no₃)₂ (aq) + nai (aq) →
options:
- nano₃ (aq) + pbi₂ (s)
- nano₃ (aq) + pbi₂ (aq)
- nano₃ (s) + pbi₂ (s)
- nano₃ (s) + pbi₂ (aq)
This is a double - displacement reaction. The reactants are $\ce{Pb(NO_{3})_{2}(aq)}$ and $\ce{NaI(aq)}$. In a double - displacement reaction, the cations and anions of the two compounds exchange places. The general form is $AB + CD=AD + CB$. Here, $\ce{Pb^{2+}}$ from $\ce{Pb(NO_{3})_{2}}$ will combine with $\ce{I^{-}}$ from $\ce{NaI}$, and $\ce{Na^{+}}$ from $\ce{NaI}$ will combine with $\ce{NO_{3}^{-}}$ from $\ce{Pb(NO_{3})_{2}}$.
The reaction equation is $\ce{Pb(NO_{3})_{2}(aq) + 2NaI(aq)=2NaNO_{3}(aq) + PbI_{2}(s)}$. We know that lead(II) iodide ($\ce{PbI_{2}}$) is insoluble in water, so it will be a solid ($\ce{(s)}$), and sodium nitrate ($\ce{NaNO_{3}}$) is soluble in water, so it will be in the aqueous state ($\ce{(aq)}$). Looking at the options, the correct one should have $\ce{NaNO_{3}(aq) + PbI_{2}(s)}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{\ce{NaNO_{3}(aq) + PbI_{2}(s)}}$ (assuming the first option has a typo and should be this, or if the first option is $\ce{NaNO_{3}(aq) + PbI_{2}(s)}$ then that is the answer)