QUESTION IMAGE
Question
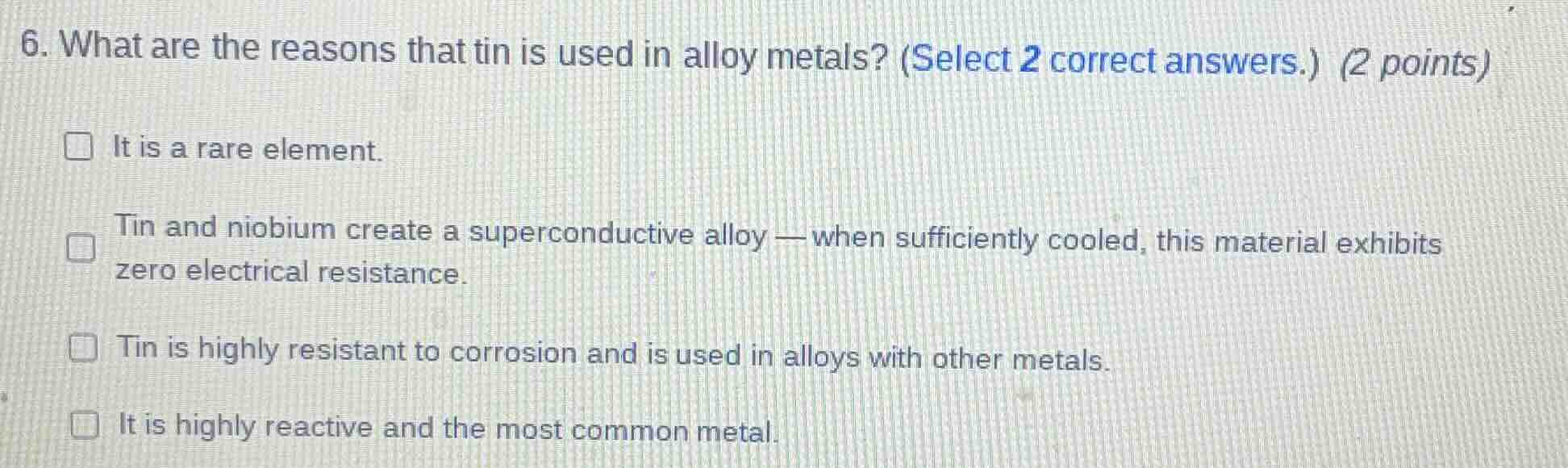
- what are the reasons that tin is used in alloy metals? (select 2 correct answers.) (2 points)
□ it is a rare element.
□ tin and niobium create a superconductive alloy — when sufficiently cooled, this material exhibits zero electrical resistance.
□ tin is highly resistant to corrosion and is used in alloys with other metals.
□ it is highly reactive and the most common metal.
Brief Explanations
- Analyze each option:
- "It is a rare element": Tin is not rare, so this is incorrect.
- "Tin and niobium create a superconductive alloy — when sufficiently cooled, this material exhibits zero electrical resistance": This is a valid reason for using tin in alloys (superconductive property).
- "Tin is highly resistant to corrosion and is used in alloys with other metals": Corrosion resistance is a key property for alloy usage, so this is valid.
- "It is highly reactive and the most common metal": Tin is not highly reactive (it's relatively stable) and not the most common metal, so this is incorrect.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. Tin and niobium create a superconductive alloy — when sufficiently cooled, this material exhibits zero electrical resistance.
C. Tin is highly resistant to corrosion and is used in alloys with other metals.