QUESTION IMAGE
Question
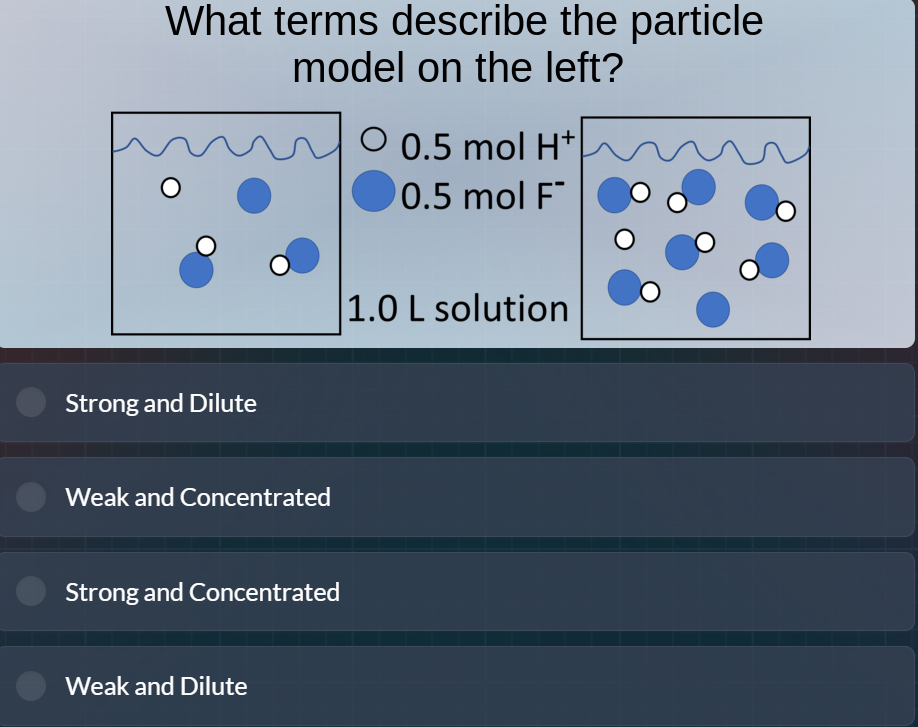
what terms describe the particle model on the left? 0.5 mol h⁺ 0.5 mol f⁻ 1.0 l solution strong and dilute weak and concentrated strong and concentrated weak and dilute
Brief Explanations
- First, analyze the dissociation: The left particle model shows some undissociated HF (since \(H^+\) and \(F^-\) are not fully dissociated, as there are bonded \(H - F\) like structures), so it's a weak acid (weak electrolyte, doesn't fully dissociate).
- Then, check concentration: Moles of \(H^+\) and \(F^-\) are 0.5 mol each in 1.0 L, so concentration is 0.5 M (dilute? Wait, no—wait, the left has fewer particles, but wait, the right has more? Wait, no, the left: total moles of solute (dissociated + undissociated). Wait, initial moles: if it's HF, initial moles would be (0.5 mol \(H^+\) + 0.5 mol \(F^-\) + undissociated HF). Wait, the left has some undissociated (the bonded \(H - F\) circles), so it's a weak acid (weak, not strong). Now, concentration: 0.5 mol \(H^+\) and 0.5 mol \(F^-\) in 1 L, but also undissociated. But the key is: weak (because it doesn't fully dissociate) and dilute? Wait, no—wait, the left has fewer particles than the right? Wait, no, the left's particle count: let's see, the left has 0.5 mol \(H^+\) (white circles), 0.5 mol \(F^-\) (blue circles), and some undissociated HF (the blue circles with white circles attached). So total solute: 0.5 + 0.5 + undissociated. But the concentration here: 0.5 M for \(H^+\) and \(F^-\), but since it's weak (partial dissociation), and the number of particles is low (dilute? Wait, no—wait, the right has more particles, so left is dilute? Wait, no, the left's solution volume is 1.0 L, same as right? Wait, the left and right are both 1.0 L? Wait, the middle says 1.0 L solution. So left is 1.0 L, with 0.5 mol \(H^+\), 0.5 mol \(F^-\), and undissociated HF. So concentration: the total solute is less than if it were strong (which would fully dissociate). But the terms: weak (because partial dissociation) and dilute (because the number of particles is low, as compared to a concentrated solution). Wait, but let's re-express:
- Strong acid: fully dissociates, so all \(H^+\) and \(A^-\) would be separate. Weak acid: partially dissociates, so some undissociated \(HA\) (like the bonded \(H - F\) here).
- Dilute: low concentration (moles per liter), concentrated: high. Here, 0.5 mol \(H^+\) and 0.5 mol \(F^-\) in 1 L, plus undissociated. But the particle density (number of particles per unit volume) is low (since there are fewer particles than a concentrated solution). So left has fewer particles, so dilute.
So the left is a weak acid (partial dissociation) and dilute (low concentration of ions/particles). So the correct option is Weak and Dilute.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. Weak and Dilute