QUESTION IMAGE
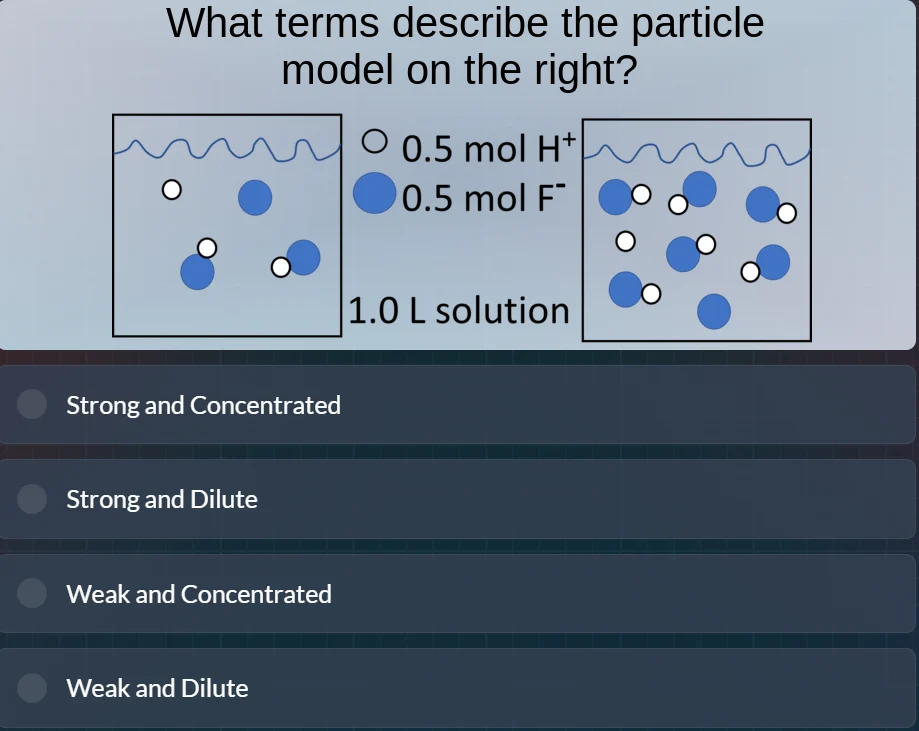
Question
what terms describe the particle model on the right?
○ 0.5 mol h⁺
● 0.5 mol f⁻
1.0 l solution
options:
strong and concentrated
strong and dilute
weak and concentrated
weak and dilute
Brief Explanations
- First, analyze the dissociation: HF (hydrofluoric acid) is a weak acid, so it doesn't dissociate completely. But looking at the particle models, the right - hand model has more particles (ions) than the left.
- Concentration: Molarity \(M=\frac{n}{V}\). For the right - hand side, if we consider the total moles of ions (assuming more dissociation or more solute), and the volume is 1.0 L. A concentrated solution has a relatively high amount of solute per unit volume. Also, since HF is weak, but the particle count in the right model is higher (more ions), so it's concentrated. And because HF is a weak acid (doesn't dissociate completely, but the concentration of solute is high here), the correct description is Weak and Concentrated.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Weak and Concentrated