QUESTION IMAGE
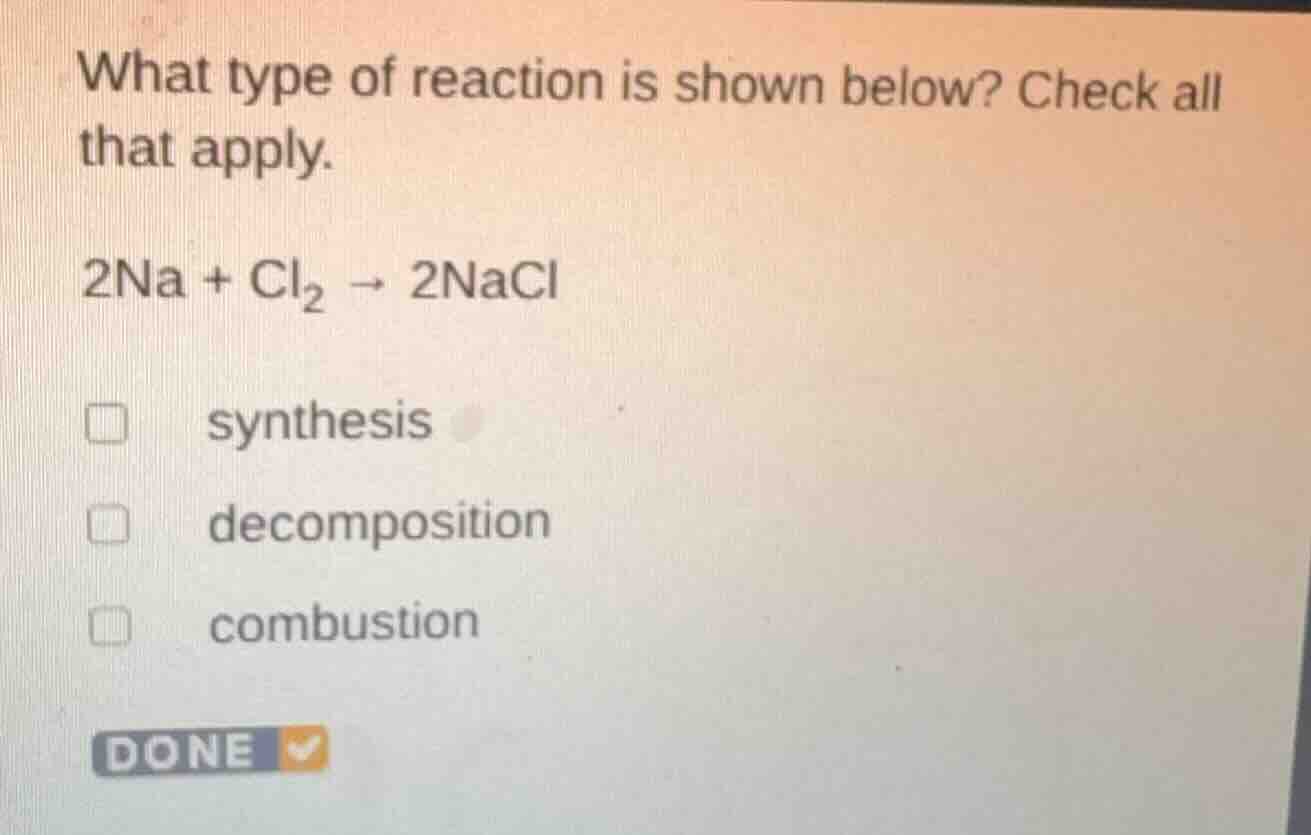
Question
what type of reaction is shown below? check all that apply.
2na + cl₂ → 2nacl
synthesis
decomposition
combustion
done
A synthesis reaction is when two or more reactants combine to form a single product. The reaction \(2\text{Na} + \text{Cl}_2
ightarrow 2\text{NaCl}\) has sodium (Na) and chlorine gas (\(\text{Cl}_2\)) as reactants combining to form sodium chloride (NaCl), so it's a synthesis reaction. Decomposition reactions involve a single compound breaking into simpler substances (not the case here). Combustion reactions typically involve a substance reacting with oxygen to produce oxides (not the case here).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. synthesis