QUESTION IMAGE
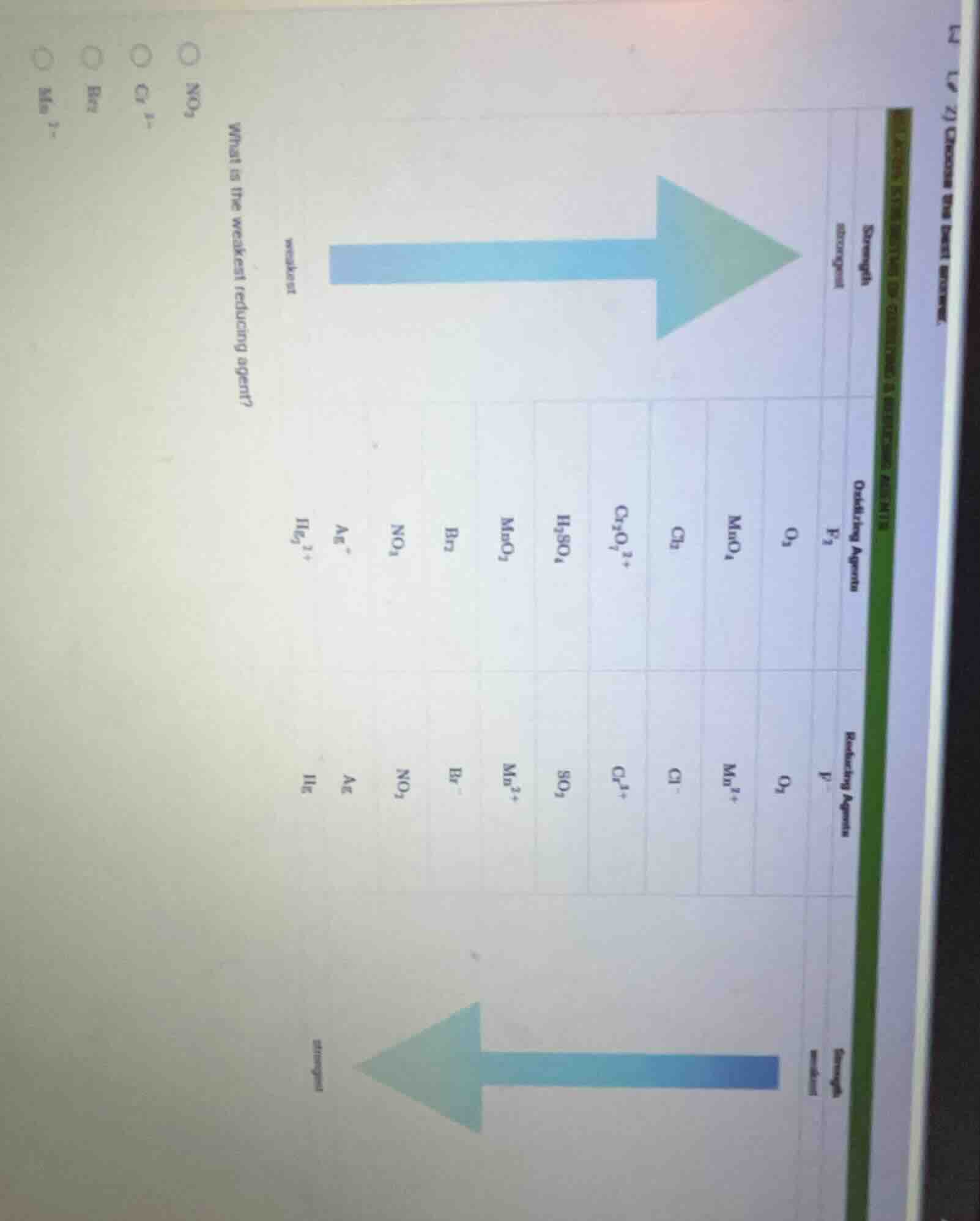
Question
what is the weakest reducing agent? \
options: $\ce{no_3^-}$, $\ce{cl^-}$, $\ce{br^-}$, $\ce{mn^{2-}}$ \
(chart showing oxidizing agents, reducing agents, and strength arrows)
The strength of reducing agents has an inverse relationship with their corresponding oxidizing agents: the strongest oxidizing agent forms the weakest reducing agent, and reducing agent strength increases in the opposite direction of oxidizing agent strength. Looking at the table, $\text{Mn}^{2+}$ is the reduced form of the strongest oxidizing agent ($\text{MnO}_4^-$), so it is the weakest reducing agent among the options.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. Mn²⁺