QUESTION IMAGE
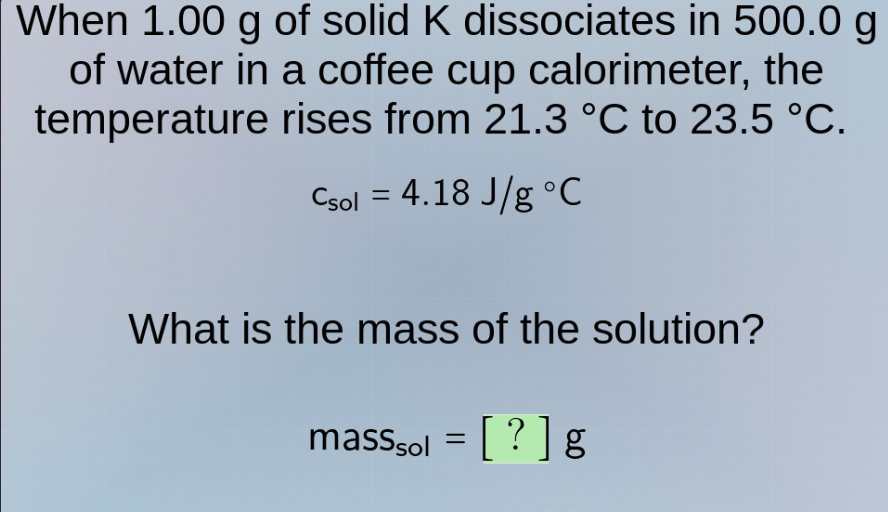
Question
when 1.00 g of solid k dissociates in 500.0 g of water in a coffee cup calorimeter, the temperature rises from 21.3 °c to 23.5 °c.
$c_{sol} = 4.18 \\, \text{j/g °c}$
what is the mass of the solution?
$\text{mass}_{sol} = ? \\, \text{g}$
Step1: Identify masses of solute and solvent
Mass of solute (K) is 1.00 g, mass of solvent (water) is 500.0 g.
Step2: Calculate mass of solution
Mass of solution = mass of solute + mass of solvent = 1.00 g + 500.0 g = 501.0 g.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
501.0