QUESTION IMAGE
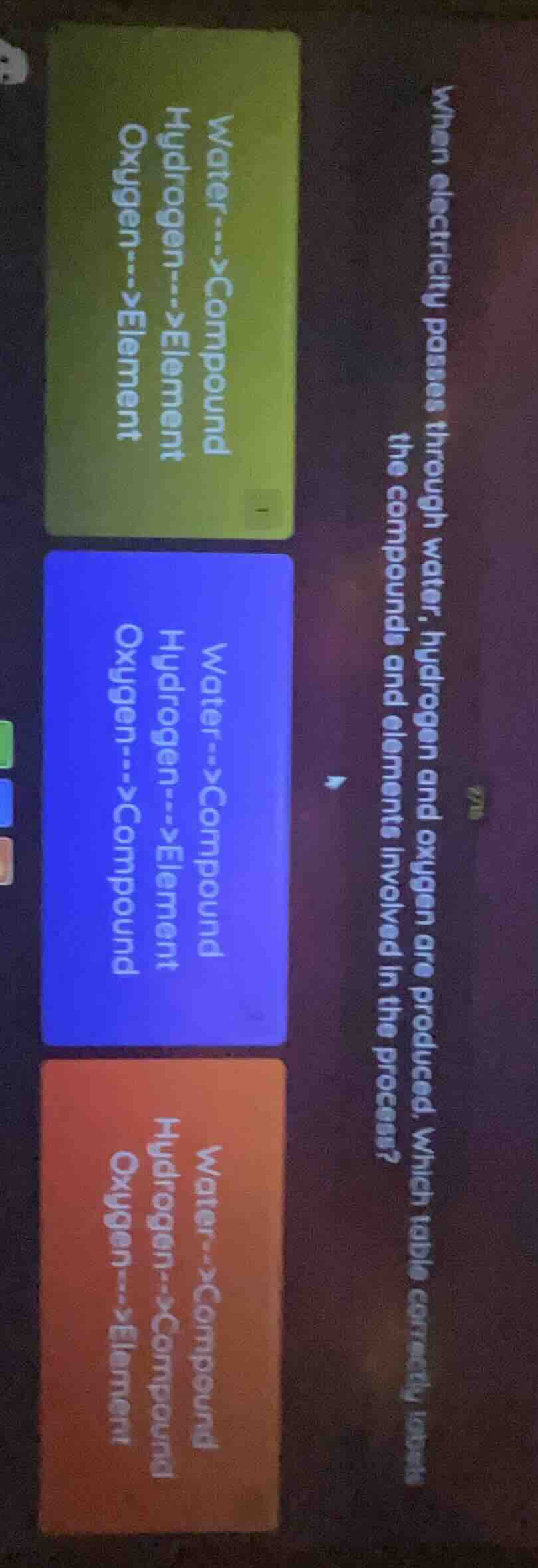
Question
when electricity passes through water, hydrogen and oxygen are produced. which table correctly labels the compounds and elements involved in the process?
water-->compound
hydrogen-->element
oxygen-->element
water-->compound
hydrogen-->element
oxygen-->compound
water-->compound
hydrogen-->compound
oxygen-->element
To solve this, we recall the definitions: A compound is a substance made of two or more elements chemically combined (e.g., water, $\ce{H_2O}$). An element is a pure substance made of only one type of atom (e.g., hydrogen, $\ce{H}$; oxygen, $\ce{O}$). During electrolysis of water, water ($\ce{H_2O}$) is the compound being decomposed. The products are hydrogen gas ($\ce{H_2}$) and oxygen gas ($\ce{O_2}$), both of which are elements (each is made of only one type of atom).
Looking at the options:
- Green option: Water (compound), Hydrogen (element), Oxygen (element) – this matches the definitions.
- Blue option: Oxygen is labeled as a compound, which is incorrect (oxygen is an element).
- Red option: Hydrogen is labeled as a compound, which is incorrect (hydrogen is an element).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The green - colored option (Water-- > Compound, Hydrogen-- > Element, Oxygen-- > Element)