QUESTION IMAGE
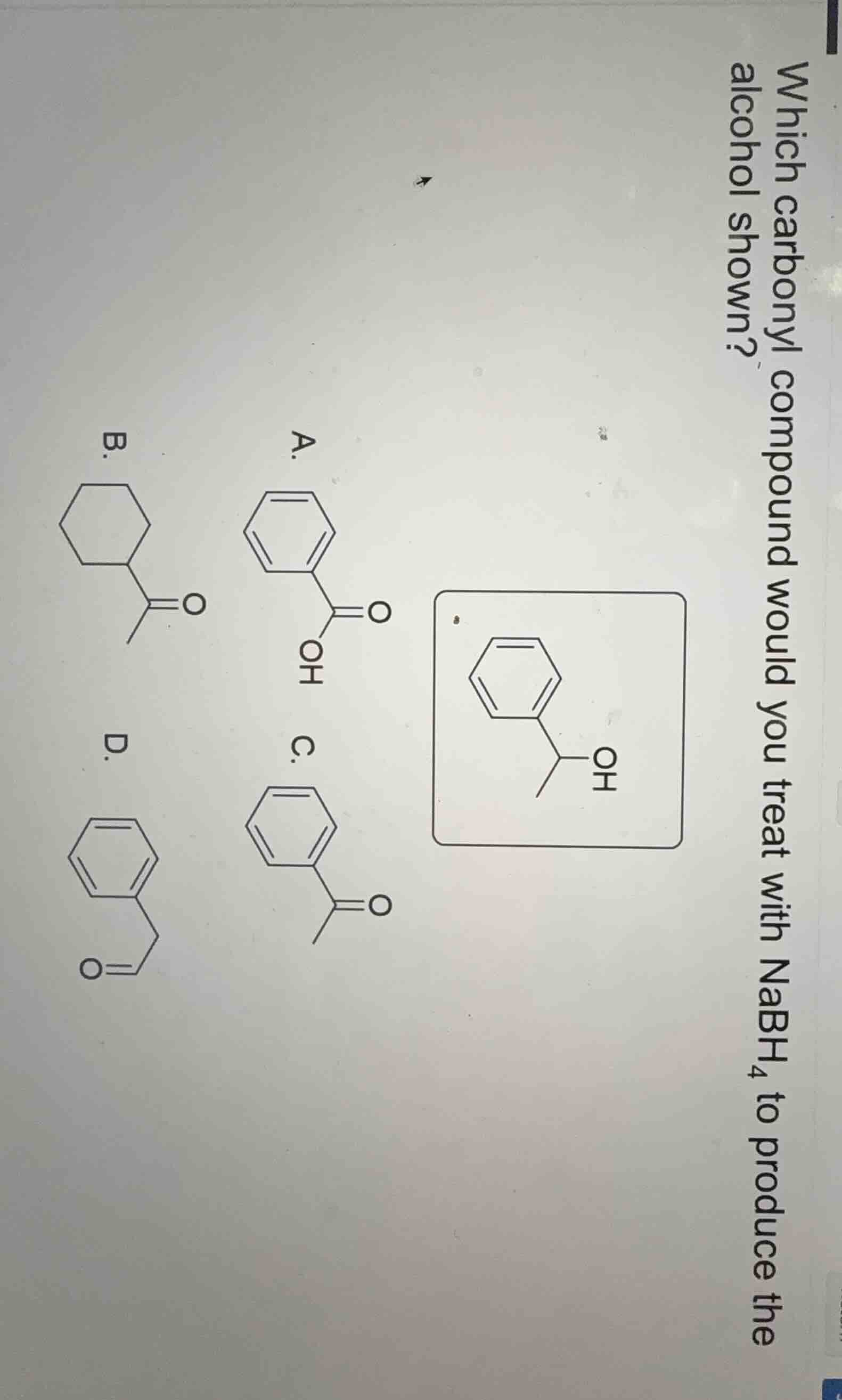
Question
which carbonyl compound would you treat with nabh₄ to produce the alcohol shown? a. chemical structure b. chemical structure c. chemical structure d. chemical structure
To determine which carbonyl compound, when treated with $\ce{NaBH_{4}}$, produces the given alcohol, we analyze the structure of the alcohol and the possible carbonyl precursors. $\ce{NaBH_{4}}$ is a reducing agent that reduces ketones to secondary alcohols and aldehydes to primary alcohols. The given alcohol has a secondary alcohol group (the $\ce{-OH}$ is attached to a carbon that is bonded to two other carbons: one from the phenyl ring and one from a methyl group, and the carbon itself is part of a structure where it was likely a ketone carbonyl).
Let's analyze each option:
- Option A: This is a carboxylic acid (has a $\ce{-COOH}$ group). $\ce{NaBH_{4}}$ does not reduce carboxylic acids (stronger reducing agents like $\ce{LiAlH_{4}}$ are needed for that). So A is incorrect.
- Option B: This has a cyclohexyl ring with a ketone group. But the product alcohol has a phenyl ring, not a cyclohexyl ring. So B is incorrect.
- Option C: This is a ketone with a phenyl ring and a methyl group attached to the carbonyl carbon (a acetophenone derivative). Reduction of a ketone (carbonyl $\ce{C=O}$) with $\ce{NaBH_{4}}$ produces a secondary alcohol. The structure of the alcohol (phenyl ring, a carbon with $\ce{-OH}$ and a methyl group) matches the product of reducing this ketone.
- Option D: This is an aldehyde (benzaldehyde derivative? Wait, no, the structure looks like a benzaldehyde? Wait, no, the structure has a formyl group? Wait, no, the D structure: let's see, it's a phenyl ring with a $\ce{-CH_{2}CHO}$? Wait, no, the D structure: the carbonyl is an aldehyde (since it's a $\ce{-CHO}$ group? Wait, no, the drawing: D has a phenyl ring and a $\ce{-CH_{2}CHO}$? Wait, no, the D structure: the carbonyl is an aldehyde, but the product alcohol is a secondary alcohol (from a ketone), not a primary alcohol (from an aldehyde). So D is an aldehyde, which would reduce to a primary alcohol, but our product is a secondary alcohol. So D is incorrect.
So the correct carbonyl compound is the ketone in option C, which upon reduction with $\ce{NaBH_{4}}$ (which reduces ketones to secondary alcohols) gives the desired alcohol.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. The ketone with a phenyl ring and a methyl group attached to the carbonyl carbon (acetophenone - type structure)