QUESTION IMAGE
Question
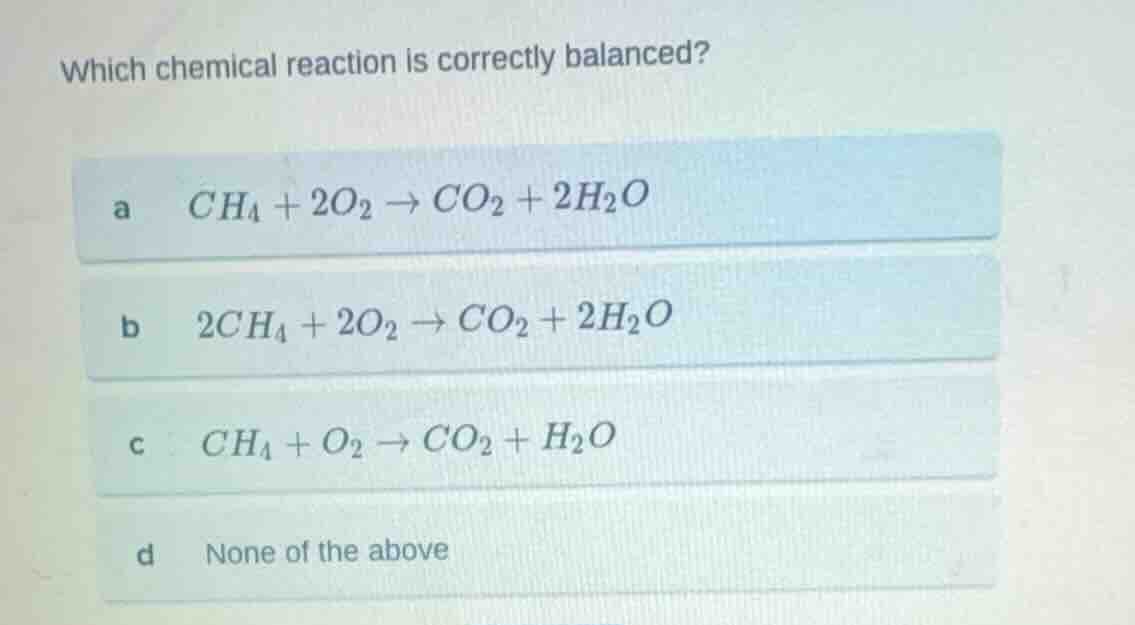
which chemical reaction is correctly balanced?
a \\( ch_4 + 2o_2 \
ightarrow co_2 + 2h_2o \\)
b \\( 2ch_4 + 2o_2 \
ightarrow co_2 + 2h_2o \\)
c \\( ch_4 + o_2 \
ightarrow co_2 + h_2o \\)
d none of the above
Brief Explanations
To determine the balanced chemical reaction, we check the number of each atom on both sides of the equation. For option a:
- Carbon (C): 1 on left ($CH_4$) and 1 on right ($CO_2$) – balanced.
- Hydrogen (H): 4 on left ($CH_4$) and \(2\times2 = 4\) on right ($2H_2O$) – balanced.
- Oxygen (O): \(2\times2 = 4\) on left ($2O_2$) and \(2 + 2\times1 = 4\) on right ($CO_2$ has 2, $2H_2O$ has 2) – balanced.
For option b:
- Carbon (C): \(2\times1 = 2\) on left ($2CH_4$) and 1 on right ($CO_2$) – unbalanced.
For option c:
- Hydrogen (H): 4 on left ($CH_4$) and 2 on right ($H_2O$) – unbalanced.
- Oxygen (O): 2 on left ($O_2$) and \(2 + 1 = 3\) on right ($CO_2$ has 2, $H_2O$ has 1) – unbalanced.
So only option a is balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a. \(CH_4 + 2O_2
ightarrow CO_2 + 2H_2O\)