QUESTION IMAGE
Question
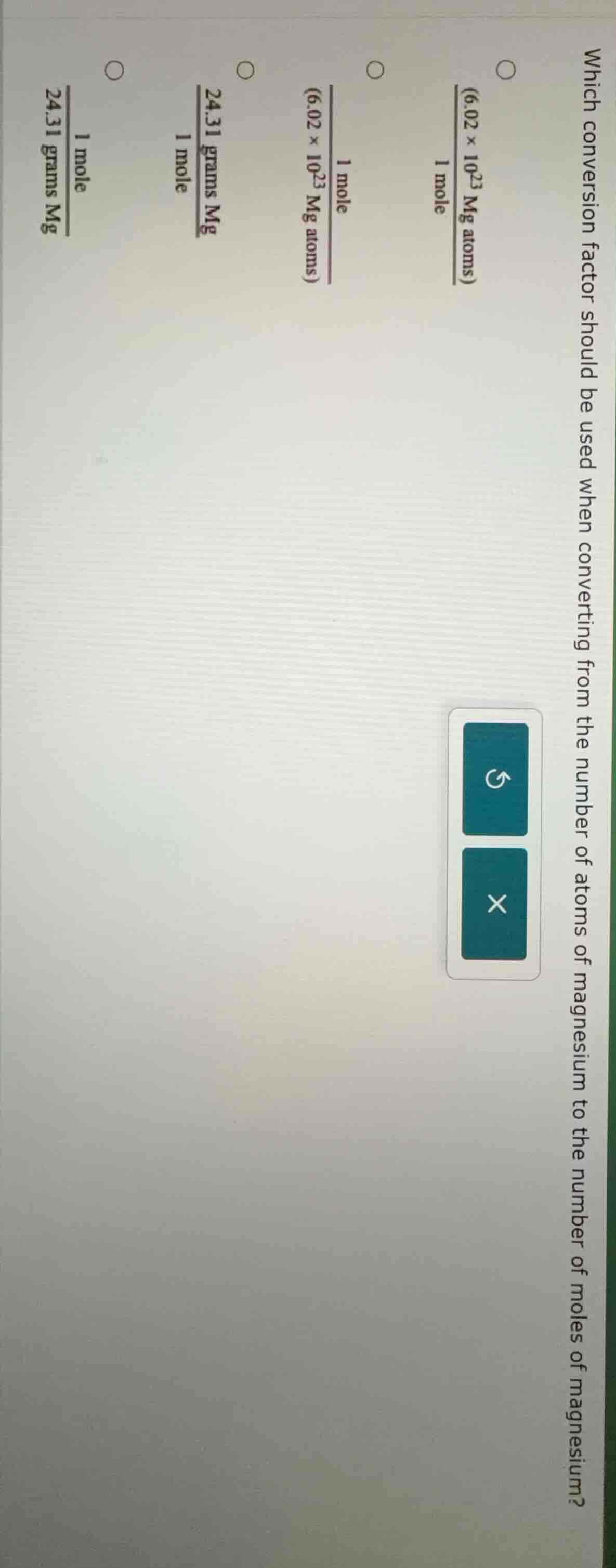
which conversion factor should be used when converting from the number of atoms of magnesium to the number of moles of magnesium? \\(\frac{(6.02 \times 10^{23}\text{ mg atoms})}{1\text{ mole}}\\) \\(\frac{1\text{ mole}}{(6.02 \times 10^{23}\text{ mg atoms})}\\) \\(\frac{24.31\text{ grams mg}}{1\text{ mole}}\\) \\(\frac{1\text{ mole}}{24.31\text{ grams mg}}\\)
Step1: Recall Avogadro's number
Avogadro's number is \(6.02\times10^{23}\) particles (atoms, molecules, etc.) per mole. So, 1 mole of Mg atoms contains \(6.02\times10^{23}\) Mg atoms.
Step2: Determine conversion factor for atoms to moles
To convert from number of atoms to moles, we use the conversion factor that relates moles and number of atoms. Since 1 mole = \(6.02\times10^{23}\) atoms, the conversion factor to go from atoms to moles is \(\frac{1\ \text{mole}}{6.02\times10^{23}\ \text{Mg atoms}}\) (or equivalently \(\frac{1\ \text{mole}}{6.02\times10^{23}\ \text{atoms of Mg}}\)). Looking at the options, the third option (let's assume the options are labeled as first: \(\frac{6.02\times 10^{23}\ \text{Mg atoms}}{1\ \text{mole}}\), second: \(\frac{1\ \text{mole}}{6.02\times 10^{23}\ \text{Mg atoms}}\), third: \(\frac{24.31\ \text{grams Mg}}{1\ \text{mole}}\), fourth: \(\frac{1\ \text{mole}}{24.31\ \text{grams Mg}}\)) – wait, re - examining the options:
Option 1: \(\frac{6.02\times 10^{23}\ \text{Mg atoms}}{1\ \text{mole}}\) (this is for converting moles to atoms)
Option 2: \(\frac{1\ \text{mole}}{6.02\times 10^{23}\ \text{Mg atoms}}\) (this is for converting atoms to moles)
Option 3: \(\frac{24.31\ \text{grams Mg}}{1\ \text{mole}}\) (this is molar mass, for mass - mole conversions)
Option 4: \(\frac{1\ \text{mole}}{24.31\ \text{grams Mg}}\) (also for mass - mole conversions, inverse of molar mass)
Since we want to convert from number of atoms to moles, we need the conversion factor with moles in the numerator and number of atoms in the denominator, which is \(\frac{1\ \text{mole}}{6.02\times 10^{23}\ \text{Mg atoms}}\) (the second option in the list of the four options, but in the given image, the third option (if we count the four options as:
- \(\frac{6.02\times 10^{23}\ \text{Mg atoms}}{1\ \text{mole}}\)
- \(\frac{1\ \text{mole}}{6.02\times 10^{23}\ \text{Mg atoms}}\)
- \(\frac{24.31\ \text{grams Mg}}{1\ \text{mole}}\)
- \(\frac{1\ \text{mole}}{24.31\ \text{grams Mg}}\)
Wait, the user's image shows four options:
First option: \(\frac{6.02\times 10^{23}\ \text{Mg atoms}}{1\ \text{mole}}\)
Second option: \(\frac{1\ \text{mole}}{6.02\times 10^{23}\ \text{Mg atoms}}\)
Third option: \(\frac{24.31\ \text{grams Mg}}{1\ \text{mole}}\)
Fourth option: \(\frac{1\ \text{mole}}{24.31\ \text{grams Mg}}\)
So to convert from number of atoms to moles, we use the conversion factor \(\frac{1\ \text{mole}}{6.02\times 10^{23}\ \text{Mg atoms}}\), which is the second option.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The conversion factor is \(\frac{1\ \text{mole}}{6.02\times 10^{23}\ \text{Mg atoms}}\) (corresponding to the option with \(\frac{1\ \text{mole}}{(6.02\times 10^{23}\ \text{Mg atoms})}\))