QUESTION IMAGE
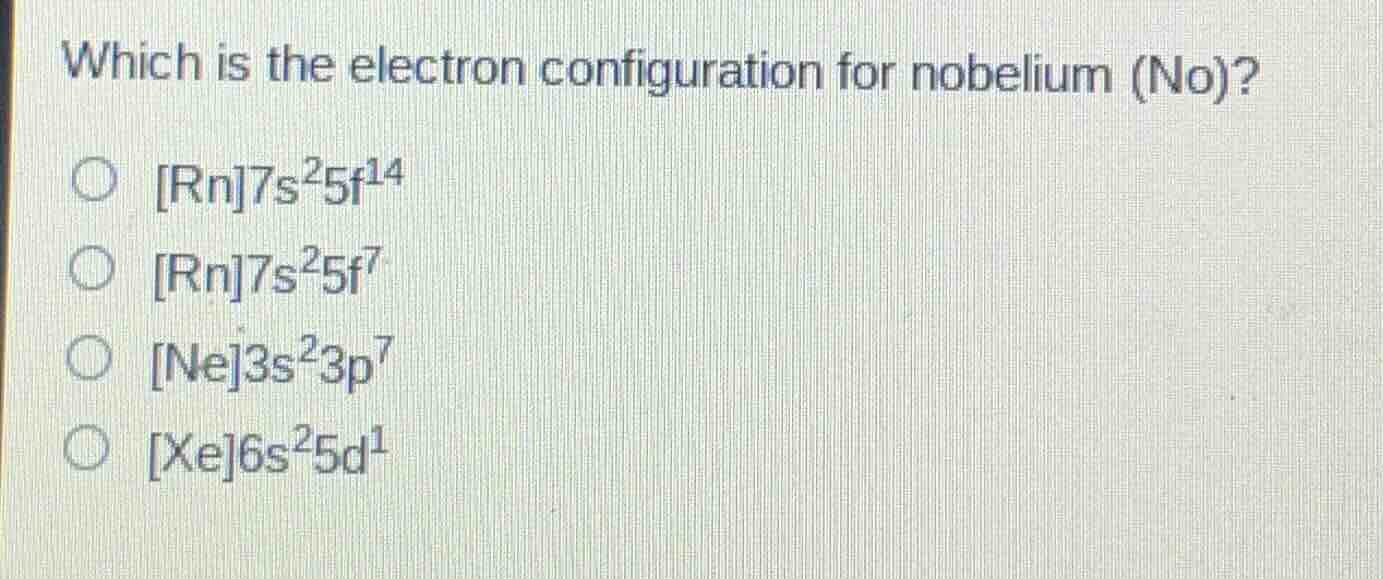
Question
which is the electron configuration for nobelium (no)?
○ rn7s²5f¹⁴
○ rn7s²5f⁷
○ ne3s²3p⁷
○ xe6s²5d¹
Brief Explanations
- First, recall the atomic number of nobelium (No), which is 102. Radon (Rn) has an atomic number of 86. So we need to account for 102 - 86 = 16 more electrons.
- Analyze each option:
- Option 1: \([Rn]7s^2 5f^{14}\). The electrons from \(7s^2\) (2 electrons) and \(5f^{14}\) (14 electrons) sum to 16, which matches the remaining electrons after Rn (86 + 2+14 = 102).
- Option 2: \([Rn]7s^2 5f^7\). 2 + 7 = 9 extra electrons, 86+9 = 95 (not 102, wrong).
- Option 3: \([Ne]3s^2 3p^7\). Ne is 10, 2 + 7 = 9, total 19 (not 102, wrong, also 3p can hold max 6 electrons).
- Option 4: \([Xe]6s^2 5d^1\). Xe is 54, 2 + 1 = 3, total 57 (not 102, wrong).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
[Rn]7s²5f¹⁴