QUESTION IMAGE
Question
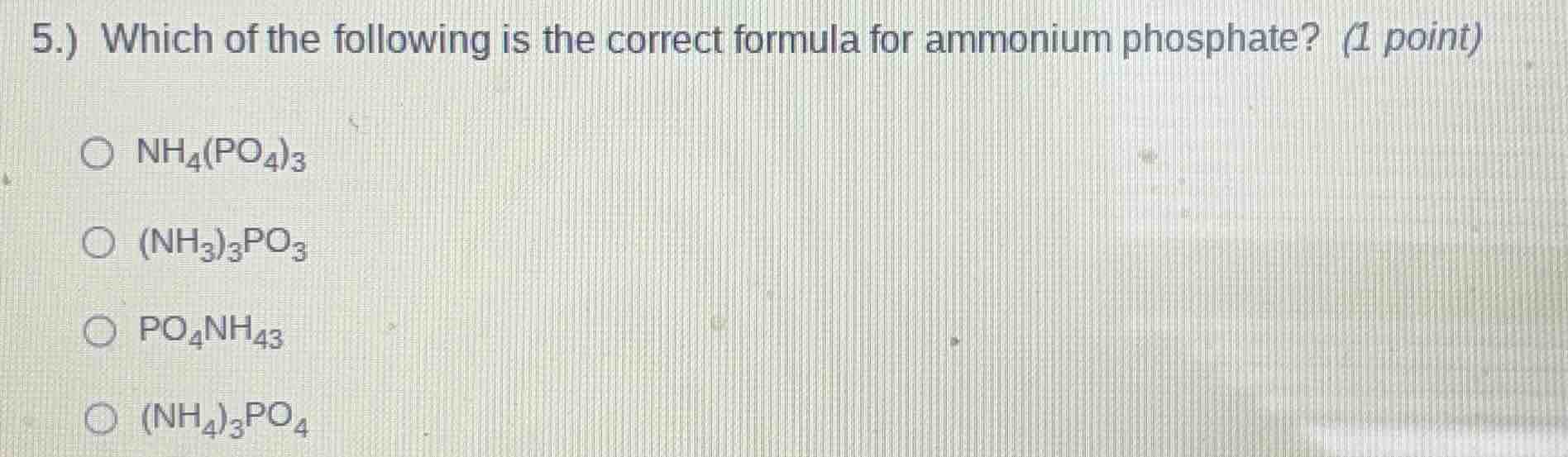
5.) which of the following is the correct formula for ammonium phosphate? (1 point) \\(\bigcirc\\) \\(\ce{nh4(po4)3}\\) \\(\bigcirc\\) \\(\ce{(nh3)3po3}\\) \\(\bigcirc\\) \\(\ce{po4nh43}\\) \\(\bigcirc\\) \\(\ce{(nh4)3po4}\\)
To determine the correct formula for ammonium phosphate, we analyze the ions involved: the ammonium ion is $\ce{NH_4^+}$ (charge +1) and the phosphate ion is $\ce{PO_4^{3-}}$ (charge -3). To balance the charges, we need 3 ammonium ions (each +1) to balance 1 phosphate ion ( - 3), so the formula is $\ce{(NH_4)_3PO_4}$. The other options have incorrect ion combinations or charges.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. $\boldsymbol{(NH_4)_3PO_4}$ (assuming the last option is labeled D; if the original options had labels like A, B, C, D with the last being D, then the answer is D. $\ce{(NH_4)_3PO_4}$)