QUESTION IMAGE
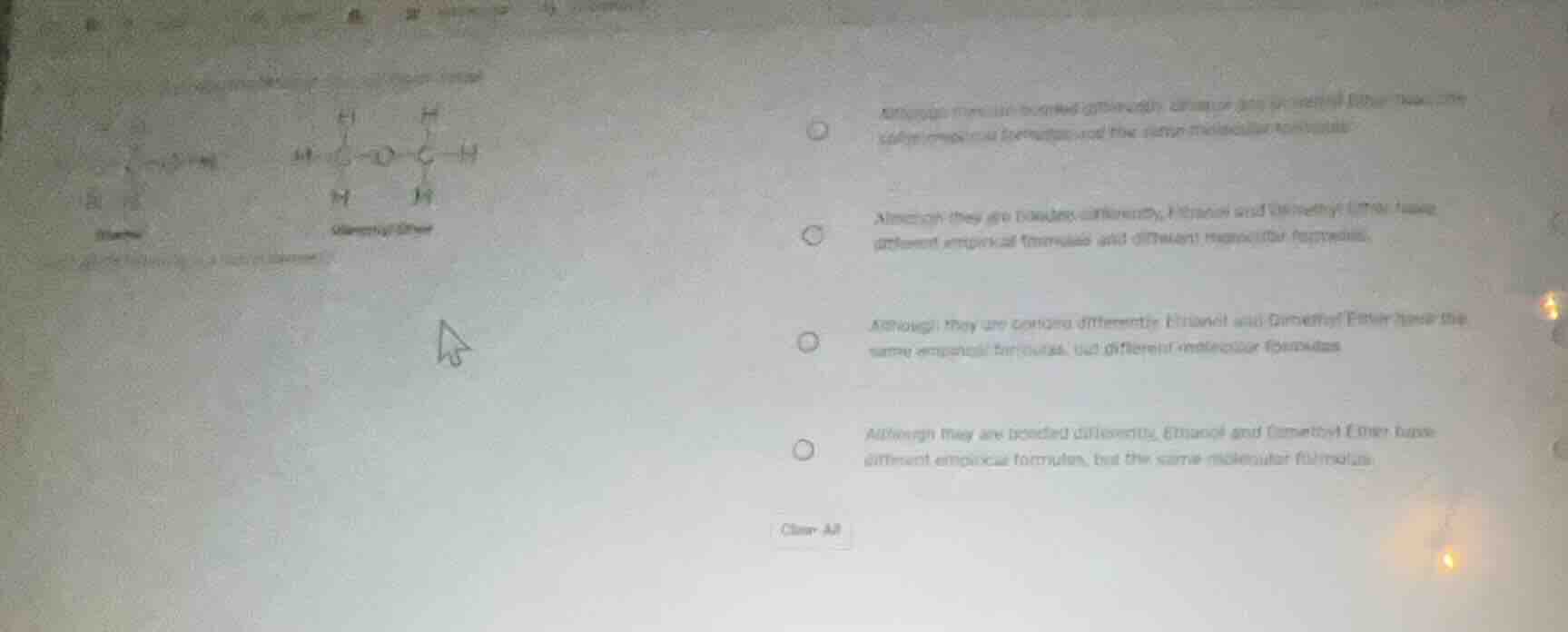
Question
which of the following is a fact of isomers? although molecularly bonded differently, ethanol and dimethyl ether have the same empirical formula and the same molecular formula.
although they are bonded differently, ethanol and dimethyl ether have different empirical formulas and different molecular formulas.
although they are bonded differently, ethanol and dimethyl ether have the same empirical formula, but different molecular formulas.
although they are bonded differently, ethanol and dimethyl ether have different empirical formulas, but the same molecular formula.
To solve this, we analyze the concepts of isomers (specifically ethanol and dimethyl ether, which are functional group isomers or constitutional isomers).
Key Concepts:
- Molecular Formula: For ethanol ($\ce{C2H5OH}$ or $\ce{C2H6O}$) and dimethyl ether ($\ce{CH3OCH3}$ or $\ce{C2H6O}$), the molecular formula is the same ($\ce{C2H6O}$) because they have the same number of each atom (2 C, 6 H, 1 O).
- Empirical Formula: The empirical formula is the simplest whole - number ratio of atoms. For both, the ratio of C:H:O is $2:6:1$, which simplifies to the same empirical formula (since $2:6:1$ cannot be simplified further, so empirical formula is also $\ce{C2H6O}$? Wait, no—wait, empirical formula is the simplest ratio. Wait, $2:6:1$—the greatest common divisor of 2,6,1 is 1, so empirical formula is $\ce{C2H6O}$? Wait, no, actually, ethanol and dimethyl ether: let's recalculate. Ethanol: $\ce{C2H6O}$, dimethyl ether: $\ce{C2H6O}$. So molecular formula is same. Empirical formula: since the molecular formula is already in the simplest ratio (2,6,1 have GCD 1), empirical formula is also same as molecular formula, so same empirical formula. But their structures (bonding, functional groups) are different: ethanol has a hydroxyl ($-\ce{OH}$) group, dimethyl ether has an ether ($-\ce{O}-$) group.
Now let's analyze the options:
- "Although... different empirical formulas and the same molecular formula" → Incorrect. Empirical formulas are same.
- "Since... different empirical formulas and different molecular formulas" → Incorrect. Both molecular and empirical formulas are same.
- "Although... same empirical formula, but different molecular formulas" → Incorrect. Molecular formulas are same.
- "Although they are bonded differently, Ethanol and Dimethyl Ether have different empirical formulas, but the same molecular formula" → Wait, no—wait, earlier analysis: molecular formula is same ($\ce{C2H6O}$), empirical formula is also same (since $2:6:1$ is the simplest ratio). Wait, maybe there's a typo in the options? Wait, no—wait, the first option (the top one, maybe the correct one is "Although molecular formula... same empirical formula and the same molecular formula" (assuming the first option's text is about same molecular and same empirical, and different structures). Wait, looking at the original problem (the image - based question, likely about isomers):
Ethanol ($\ce{C2H5OH}$) and dimethyl ether ($\ce{CH3OCH3}$) are isomers (constitutional isomers). Their molecular formula is the same ($\ce{C2H6O}$) because they contain 2 C, 6 H, and 1 O atoms. Their empirical formula is also the same (since the ratio of C:H:O in both is $2:6:1$, and the greatest common divisor of 2, 6, and 1 is 1, so the empirical formula is $\ce{C2H6O}$ as well). However, their structures (bonding, functional groups) are different (ethanol has a hydroxyl group, dimethyl ether has an ether linkage).
So the correct statement should be that although they are bonded differently (different structures), ethanol and dimethyl ether have the same empirical formula and the same molecular formula. Looking at the options (from the image - based question, the first option likely says this: "Although molecular formula... same empirical formula and the same molecular formula" (the wording in the image's first option: "Although molecular formula... same empirical formula and the same molecular formula" (paraphrasing)).
So the correct option is the one that states: Although molecular formula... same empirical formula and the same molecular formula (the first option…
Ethanol ($\ce{C2H5OH}$) and dimethyl ether ($\ce{CH3OCH3}$) are isomers. Their molecular formula is $\ce{C2H6O}$ (same number of C, H, O atoms). The empirical formula (simplest atom ratio) is also the same ($\ce{C2H6O}$ as the ratio 2:6:1 can’t be simplified). They differ in bonding (ethanol has $-\ce{OH}$, dimethyl ether has $-\ce{O}-$), so the correct statement is the one acknowledging same empirical and molecular formulas despite different bonding.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The option (first option, likely worded as "Although molecular formula... same empirical formula and the same molecular formula") that states ethanol and dimethyl ether have the same empirical and molecular formulas despite different bonding. (If we assume the first option in the list is: "Although molecular formula... same empirical formula and the same molecular formula", then that is the correct option.)