QUESTION IMAGE
Question
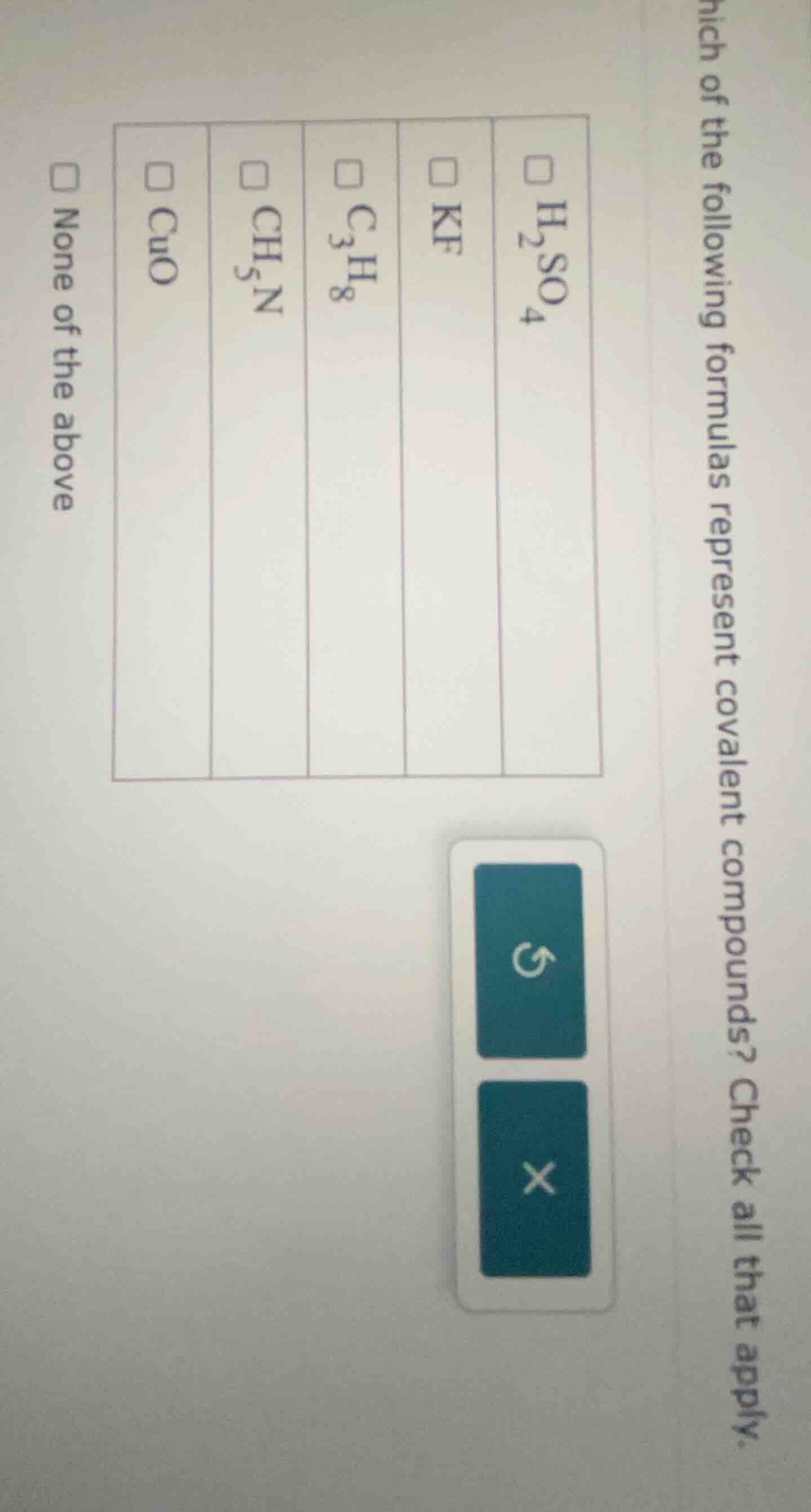
which of the following formulas represent covalent compounds? check all that apply.
□ $\ce{h_{2}so_{4}}$
□ $\ce{kf}$
□ $\ce{c_{3}h_{8}}$
□ $\ce{ch_{5}n}$
□ $\ce{cuo}$
□ none of the above
To determine covalent compounds, we identify those formed by non - metal - non - metal bonding (or with a metalloid in a covalent - like bond).
- $H_2SO_4$: Sulfuric acid has covalent bonds within the $SO_4^{2 - }$ group and polar covalent bonds with $H^+$. But actually, $H_2SO_4$ has both covalent and ionic - like interactions, but in terms of the structure of the molecule, the bonds between S, O and O, H are covalent. However, a better way is to look at the type of elements.
- $KF$: Potassium fluoride is an ionic compound (metal - non - metal, K is metal, F is non - metal).
- $C_3H_8$ (propane): It is a hydrocarbon, formed by carbon and hydrogen (both non - metals) with covalent bonds.
- $CH_5N$: This is a compound with C, H, N (all non - metals) bonded covalently.
- $CuO$: Copper(II) oxide is an ionic compound (metal - non - metal, Cu is metal, O is non - metal).
So the covalent compounds are $C_3H_8$ and $CH_5N$. Wait, actually, $H_2SO_4$: Let's re - evaluate. The bonds in $H_2SO_4$: The S - O and O - H bonds are covalent. But $H_2SO_4$ can dissociate into ions, but the molecule itself has covalent bonds. However, the standard covalent compounds here are the organic - like (hydrocarbon and amine - like) and also $H_2SO_4$? Wait, no. Let's recall: Covalent compounds are usually formed between non - metals (or metalloids and non - metals) and have a molecular structure. Ionic compounds are formed between metals and non - metals (or polyatomic ions with metal cations).
- $H_2SO_4$: Sulfur is a non - metal, oxygen is a non - metal, hydrogen is a non - metal. But $H_2SO_4$ is a molecular compound with covalent bonds.
- $KF$: Ionic (K⁺ and F⁻).
- $C_3H_8$: Covalent (C and H are non - metals, covalent bonds).
- $CH_5N$: Covalent (C, H, N are non - metals, covalent bonds).
- $CuO$: Ionic (Cu²⁺ and O²⁻).
Wait, but maybe the intended answer is $C_3H_8$ (propane, a hydrocarbon, covalent), $CH_5N$ (methylamine, covalent), and maybe $H_2SO_4$? Wait, no, $H_2SO_4$ has some ionic character in solution but the molecule is covalent. But let's check the elements:
- $H_2SO_4$: H (non - metal), S (non - metal), O (non - metal) → covalent bonds in the molecule.
- $KF$: K (metal), F (non - metal) → ionic.
- $C_3H_8$: C (non - metal), H (non - metal) → covalent.
- $CH_5N$: C (non - metal), H (non - metal), N (non - metal) → covalent.
- $CuO$: Cu (metal), O (non - metal) → ionic.
So the covalent compounds are $H_2SO_4$, $C_3H_8$, $CH_5N$? Wait, no, $H_2SO_4$: The structure of $H_2SO_4$ is a molecular compound with covalent bonds. But maybe the question considers $H_2SO_4$ as covalent? Wait, let's check each:
- $H_2SO_4$: Contains covalent bonds (S - O, O - H), so it's a covalent compound (molecular).
- $KF$: Ionic (metal - non - metal).
- $C_3H_8$: Covalent (hydrocarbon, all non - metal elements bonded covalently).
- $CH_5N$: Covalent (organic compound, non - metals bonded covalently).
- $CuO$: Ionic (metal - non - metal).
But maybe the correct ones are $C_3H_8$, $CH_5N$, and $H_2SO_4$? Wait, no, let's recall the definition of covalent compounds: Compounds where atoms are held together by covalent bonds (sharing of electrons), usually formed between non - metals. Ionic compounds are held together by ionic bonds (transfer of electrons, electrostatic attraction between ions), formed between metals and non - metals (or polyatomic ions with metal cations).
So:
- $H_2SO_4$: Covalent (non - metals bonded).
- $KF$: Ionic (K is metal, F is non - metal).
- $C_3H_8$: Covalent (C and H are non - metals).
- $CH_5N$: Covalent (C, H, N are non - metals)…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $H_2SO_4$: Covalent compound (formed by non - metals with covalent bonds).
- $C_3H_8$: Covalent compound (hydrocarbon, non - metal atoms bonded covalently).
- $CH_5N$: Covalent compound (non - metal atoms bonded covalently).
So the correct options to check are $H_2SO_4$, $C_3H_8$, $CH_5N$. But wait, maybe the intended answer is $C_3H_8$ and $CH_5N$ (since $H_2SO_4$ is a strong acid and sometimes considered to have ionic character in solution, but the molecule is covalent). If we follow the strict non - metal - non - metal bonding for covalent, then $C_3H_8$ (C and H), $CH_5N$ (C, H, N), and $H_2SO_4$ (H, S, O) are covalent. So the answer is to check $H_2SO_4$, $C_3H_8$, $CH_5N$.