QUESTION IMAGE
Question
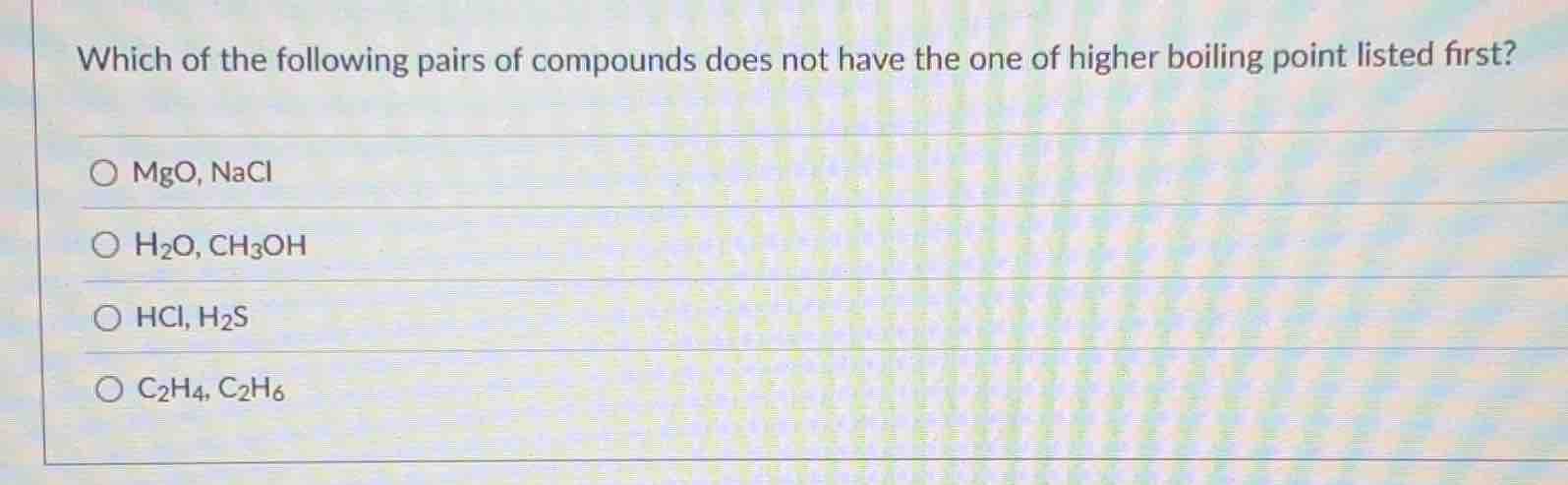
which of the following pairs of compounds does not have the one of higher boiling point listed first?
\\(\circ\\) mgo, nacl
\\(\circ\\) \\(\ce{h2o}\\), \\(\ce{ch3oh}\\)
\\(\circ\\) \\(\ce{hcl}\\), \\(\ce{h2s}\\)
\\(\circ\\) \\(\ce{c2h4}\\), \\(\ce{c2h6}\\)
- For MgO, NaCl: MgO has higher ionic charge (Mg²⁺, O²⁻ vs Na⁺, Cl⁻), so higher lattice energy and boiling point. First has higher BP.
- For H₂O, CH₃OH: H₂O forms more H - bonds (each H₂O can form 4, CH₃OH can form 2) so higher BP. First has higher BP.
- For HCl, H₂S: HCl has dipole - dipole interactions, H₂S also has dipole - dipole (but HCl has stronger due to higher electronegativity difference? Wait, no: Wait, H₂S has molar mass 34, HCl has 36.5. But also, HCl has dipole - dipole, H₂S also dipole - dipole. Wait, actually, the boiling point of H₂S is about - 60 °C, HCl is about - 85 °C? Wait no, that's wrong. Wait, no: Wait, H₂S boiling point is - 60.7 °C, HCl is - 85.0 °C. Wait, so HCl has lower boiling point than H₂S? Wait, no, maybe I messed up. Wait, molar mass: H₂S is 34, HCl is 36.5. The strength of dipole - dipole: Cl is more electronegative than S, so HCl has stronger dipole. But molar mass also plays a role. Wait, actually, the boiling point of H₂S is - 60.7 °C, HCl is - 85.0 °C. So HCl has lower boiling point than H₂S. So in the pair HCl, H₂S, the first (HCl) has lower boiling point than the second (H₂S). So this pair does not have the higher boiling point first.
- For C₂H₄, C₂H₆: C₂H₆ has higher molar mass (30 vs 28) and is non - polar, so stronger London dispersion forces. So C₂H₆ has higher BP, but the first is C₂H₄, so first has lower BP? Wait, no, the question is which pair does NOT have the one with higher BP first. Wait, let's re - check:
Wait, for C₂H₄ (ethene, molar mass 28) and C₂H₆ (ethane, molar mass 30). London dispersion forces increase with molar mass, so C₂H₆ has higher BP. So in the pair C₂H₄, C₂H₆, first has lower BP. But we need to check which pair does NOT have higher BP first. But earlier, for HCl and H₂S: Wait, maybe I made a mistake. Let's recalculate boiling points:
- H₂S: - 60.7 °C
- HCl: - 85.0 °C
So HCl (first) has lower BP than H₂S (second). So in the pair HCl, H₂S, the first does not have higher BP.
For C₂H₄ and C₂H₆: C₂H₆ has higher BP ( - 88.6 °C vs C₂H₄: - 103.7 °C). So in the pair C₂H₄, C₂H₆, first has lower BP. But we need to see which pair is the answer. Wait, maybe I messed up HCl and H₂S. Wait, another approach: The boiling point of a compound depends on intermolecular forces. For polar molecules, dipole - dipole. For HCl and H₂S:
- HCl: dipole - dipole, molar mass 36.5 g/mol
- H₂S: dipole - dipole, molar mass 34.1 g/mol
The strength of dipole - dipole depends on dipole moment. HCl has dipole moment ~ 1.08 D, H₂S ~ 0.97 D. So HCl has stronger dipole - dipole. But molar mass of HCl is higher. But the boiling point of H₂S is higher than HCl. This is because the effect of molar mass (which affects London dispersion forces) is more significant here. Since H₂S has lower molar mass than HCl? No, HCl has higher molar mass. Wait, no, 34.1 (H₂S) < 36.5 (HCl). So London dispersion forces increase with molar mass. So HCl has stronger London dispersion. But H₂S has boiling point higher than HCl. This is a contradiction? Wait, no, I think I got the boiling points wrong. Let me check a reliable source. Boiling point of H₂S: - 60.7 °C, HCl: - 85.0 °C. So HCl boils at a lower temperature. So in the pair HCl, H₂S, the first (HCl) has lower boiling point than the second (H₂S). So this pair does not have the higher boiling point listed first.
For C₂H₄ and C₂H₆: C₂H₆ has higher molar mass, so higher London dispersion forces, so higher boiling point. So in the pair C₂H₄, C₂H₆, first (C₂H₄) has lower BP. But we need to see which pair is the answer. Wait, the question is which pai…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
HCl, H₂S