QUESTION IMAGE
Question
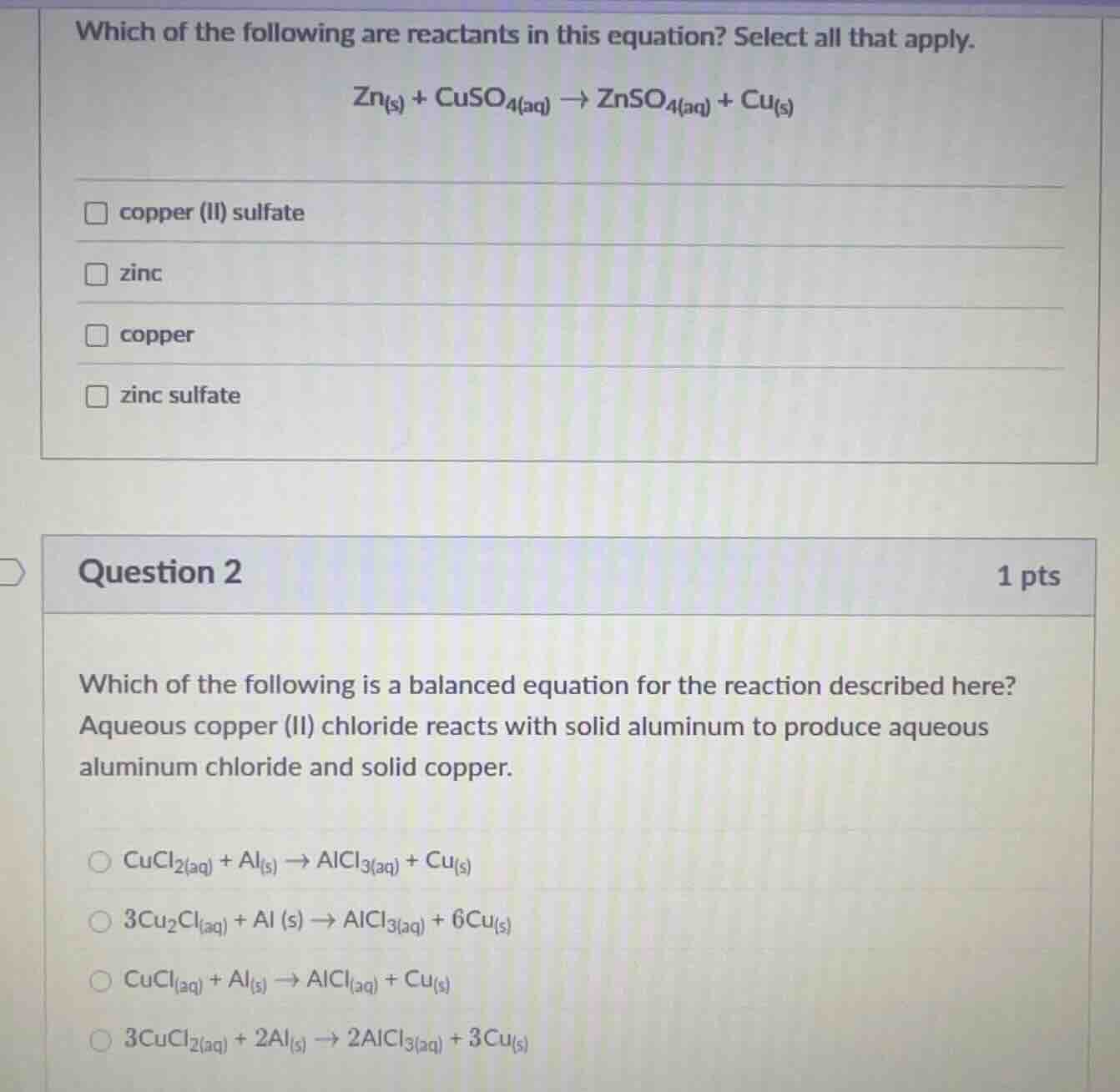
which of the following are reactants in this equation? select all that apply.
zn₍ₛ₎ + cuso₄₍ₐq₎ → znso₄₍ₐq₎ + cu₍ₛ₎
□ copper (ii) sulfate
□ zinc
□ copper
□ zinc sulfate
question 2
1 pts
which of the following is a balanced equation for the reaction described here?
aqueous copper (ii) chloride reacts with solid aluminum to produce aqueous
aluminum chloride and solid copper.
○ cucl₂₍ₐq₎ + al₍ₛ₎ → alcl₃₍ₐq₎ + cu₍ₛ₎
○ 3cu₂cl₍ₐq₎ + al (s) → alcl₃₍ₐq₎ + 6cu₍ₛ₎
○ cucl₍ₐq₎ + al₍ₛ₎ → alcl₃₍ₐq₎ + cu₍ₛ₎
○ 3cucl₂₍ₐq₎ + 2al₍ₛ₎ → 2alcl₃₍ₐq₎ + 3cu₍ₛ₎
(First Question):
Step1: Identify reactant side
In a chemical equation $\text{Reactants}
ightarrow \text{Products}$, the left side of the arrow are reactants: $\text{Zn}_{(s)} + \text{CuSO}_{4(aq)}$
Step2: Match to names
$\text{Zn}$ is zinc; $\text{CuSO}_4$ is copper (II) sulfate.
(Second Question):
Step1: Write unbalanced equation
Based on description: $\text{CuCl}_{2(aq)} + \text{Al}_{(s)}
ightarrow \text{AlCl}_{3(aq)} + \text{Cu}_{(s)}$
Step2: Balance Cl atoms
Add coefficients to balance Cl: $3\text{CuCl}_{2(aq)} + \text{Al}_{(s)}
ightarrow 2\text{AlCl}_{3(aq)} + \text{Cu}_{(s)}$
Step3: Balance Cu and Al atoms
Adjust coefficients for Cu and Al: $3\text{CuCl}_{2(aq)} + 2\text{Al}_{(s)}
ightarrow 2\text{AlCl}_{3(aq)} + 3\text{Cu}_{(s)}$
Step4: Verify atom counts
Left: 3 Cu, 6 Cl, 2 Al; Right: 2 Al, 6 Cl, 3 Cu. All atoms balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
First question (select all that apply):
- copper (II) sulfate
- zinc
Second question:
3CuCl₂(aq) + 2Al(s) → 2AlCl₃(aq) + 3Cu(s)