QUESTION IMAGE
Question
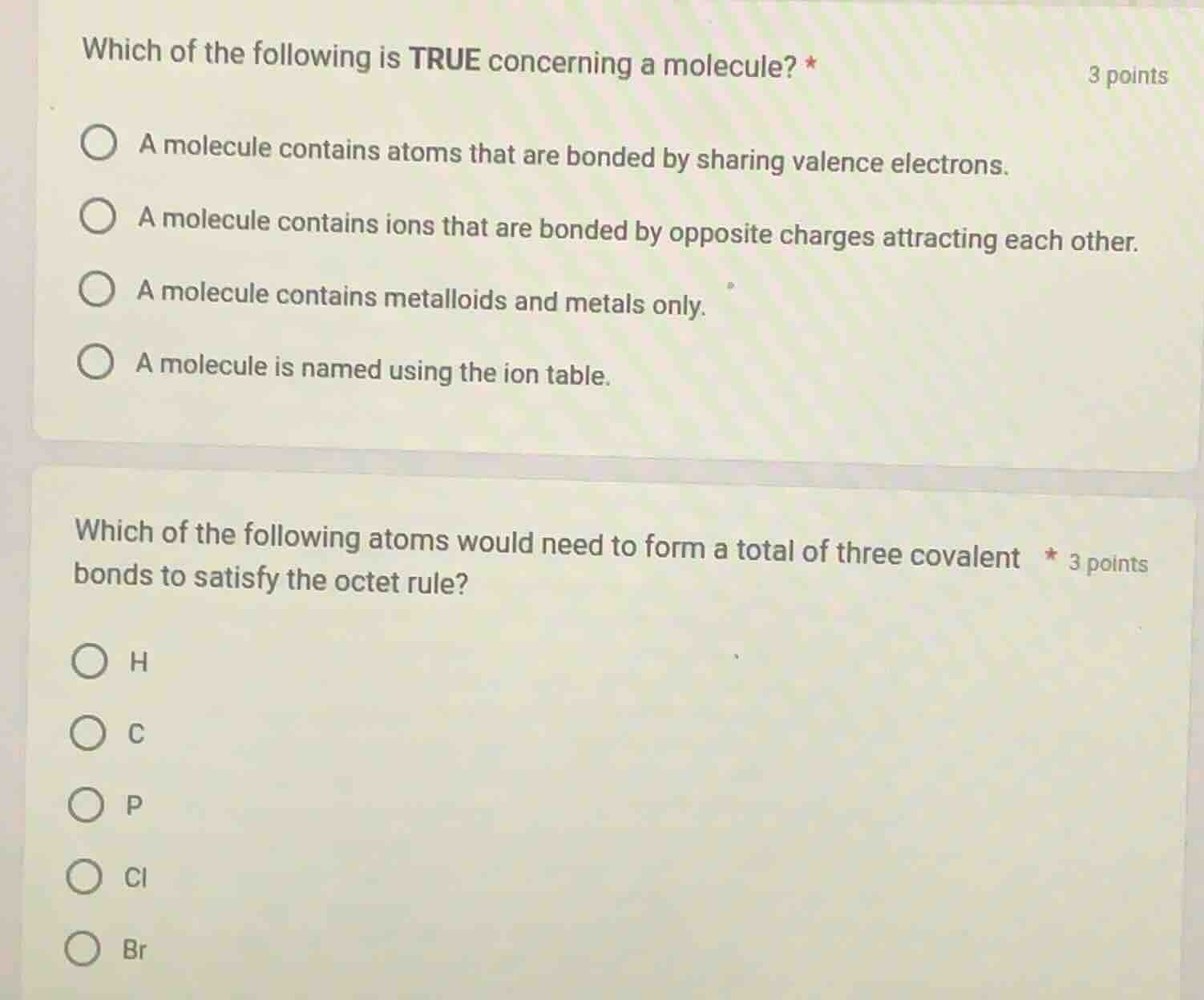
which of the following is true concerning a molecule? *
a molecule contains atoms that are bonded by sharing valence electrons.
a molecule contains ions that are bonded by opposite charges attracting each other.
a molecule contains metalloids and metals only.
a molecule is named using the ion table.
which of the following atoms would need to form a total of three covalent bonds to satisfy the octet rule?
h
c
p
cl
br
Brief Explanations
For the first question:
- Molecules are formed by covalent bonding, which involves sharing valence electrons. Ionic bonds (opposite charge attraction) form ionic compounds, not molecules. Molecules can include nonmetals, not just metalloids/metals. Ion tables are used for ionic compounds, not molecules.
For the second question:
- Hydrogen (H) only needs 2 electrons (duet rule) and forms 1 bond. Carbon (C) has 4 valence electrons, needs 4 bonds to reach 8. Phosphorus (P) has 5 valence electrons; forming 3 covalent bonds adds 3 electrons, giving it 8 total (satisfies the octet rule). Chlorine (Cl) and Bromine (Br) have 7 valence electrons each, needing only 1 bond to reach 8.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- A molecule contains atoms that are bonded by sharing valence electrons.
- P