QUESTION IMAGE
Question
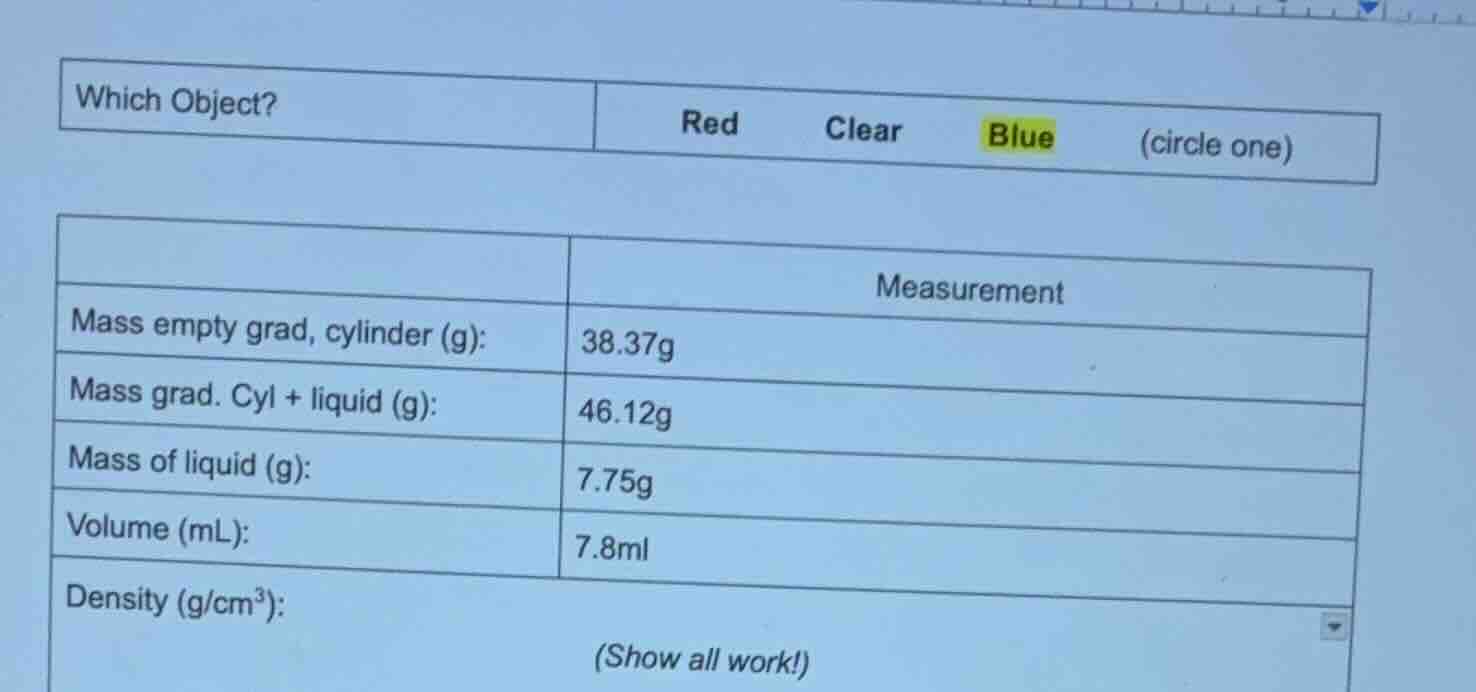
which object? \tred \tclear \tblue \t(circle one)
\t\tmeasurement
mass empty grad, cylinder (g):\t38.37g
mass grad. cyl + liquid (g):\t46.12g
mass of liquid (g):\t7.75g
volume (ml):\t7.8ml
density (g/cm³):
(show all work!)
Step1: Recall density formula
Density ($
ho$) is calculated by the formula $
ho = \frac{m}{V}$, where $m$ is the mass of the substance and $V$ is its volume.
Step2: Substitute values
Here, the mass of the liquid ($m$) is $7.75$ g and the volume ($V$) is $7.8$ mL (since $1$ mL = $1$ $cm^3$, the volume in $cm^3$ is also $7.8$). Substitute into the formula: $
ho = \frac{7.75\ g}{7.8\ cm^3}$.
Step3: Calculate the density
Perform the division: $\frac{7.75}{7.8} \approx 0.994\ g/cm^3$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The density of the liquid is approximately $\boldsymbol{0.994\ g/cm^3}$ (or more precisely, if we keep more decimal places during calculation, $\frac{7.75}{7.8}\approx0.99359\approx0.99\ g/cm^3$ depending on significant figures, but with the given values, the calculation as above gives approximately $0.994\ g/cm^3$).