QUESTION IMAGE
Question
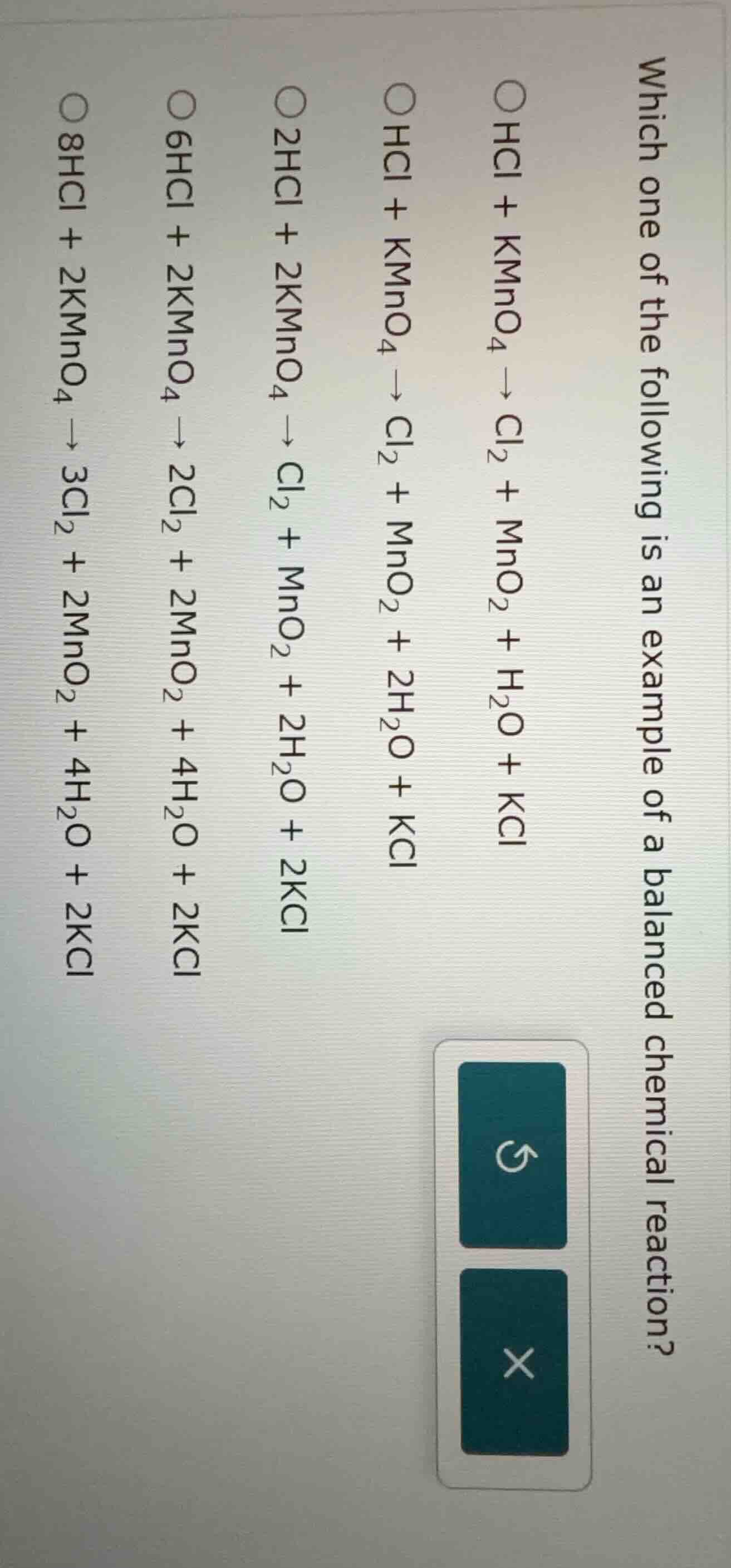
which one of the following is an example of a balanced chemical reaction?
- hcl + kmno₄ → cl₂ + mno₂ + h₂o + kcl
- hcl + kmno₄ → cl₂ + mno₂ + 2h₂o + kcl
- 2hcl + 2kmno₄ → cl₂ + mno₂ + 2h₂o + 2kcl
- 6hcl + 2kmno₄ → 2cl₂ + 2mno₂ + 4h₂o + 2kcl
- 8hcl + 2kmno₄ → 3cl₂ + 2mno₂ + 4h₂o + 2kcl
To determine a balanced chemical reaction, we check the number of each atom on both sides (reactants and products).
Step1: Analyze Option 1 ($\ce{HCl + KMnO4 -> Cl2 + MnO2 + H2O + KCl}$)
- H: 1 on left, 2 on right (unbalanced)
- Cl: 1 on left, 3 (2 in $\ce{Cl2}$ + 1 in $\ce{KCl}$) on right (unbalanced)
- Mn: 1 on left, 1 on right (balanced)
- K: 1 on left, 1 on right (balanced)
- O: 4 on left, 3 (2 in $\ce{MnO2}$ + 1 in $\ce{H2O}$) on right (unbalanced)
Step2: Analyze Option 2 ($\ce{HCl + KMnO4 -> Cl2 + MnO2 + 2H2O + KCl}$)
- H: 1 on left, 4 (2×2 in $\ce{H2O}$) on right (unbalanced)
- Cl: 1 on left, 3 (2 in $\ce{Cl2}$ + 1 in $\ce{KCl}$) on right (unbalanced)
- Mn: 1 on left, 1 on right (balanced)
- K: 1 on left, 1 on right (balanced)
- O: 4 on left, 5 (2 in $\ce{MnO2}$ + 2×1 in $\ce{H2O}$) on right (unbalanced)
Step3: Analyze Option 3 ($\ce{2HCl + 2KMnO4 -> Cl2 + MnO2 + 2H2O + 2KCl}$)
- H: 2 on left, 4 (2×2 in $\ce{H2O}$) on right (unbalanced)
- Cl: 2 on left, 4 (2 in $\ce{Cl2}$ + 2 in $\ce{2KCl}$) on right (unbalanced)
- Mn: 2 on left, 1 on right (unbalanced)
- K: 2 on left, 2 on right (balanced)
- O: 8 (2×4 in $\ce{2KMnO4}$) on left, 6 (2 in $\ce{MnO2}$ + 2×1 in $\ce{2H2O}$) on right (unbalanced)
Step4: Analyze Option 4 ($\ce{6HCl + 2KMnO4 -> 2Cl2 + 2MnO2 + 4H2O + 2KCl}$)
- H: 6 on left, 8 (4×2 in $\ce{4H2O}$) on right (unbalanced)
- Cl: 6 on left, 6 (2×2 in $\ce{2Cl2}$ + 2 in $\ce{2KCl}$) on right (balanced)
- Mn: 2 on left, 2 on right (balanced)
- K: 2 on left, 2 on right (balanced)
- O: 8 (2×4 in $\ce{2KMnO4}$) on left, 8 (2×2 in $\ce{2MnO2}$ + 4×1 in $\ce{4H2O}$) on right (balanced) Wait, H is unbalanced (6 vs 8).
Step5: Analyze Option 5 ($\ce{8HCl + 2KMnO4 -> 3Cl2 + 2MnO2 + 4H2O + 2KCl}$)
- H: 8 on left, 8 (4×2 in $\ce{4H2O}$) on right (balanced)
- Cl: 8 on left, 8 (3×2 in $\ce{3Cl2}$ + 2 in $\ce{2KCl}$) on right (balanced: \(3\times2 + 2 = 8\))
- Mn: 2 on left, 2 on right (balanced)
- K: 2 on left, 2 on right (balanced)
- O: 8 (2×4 in $\ce{2KMnO4}$) on left, 8 (2×2 in $\ce{2MnO2}$ + 4×1 in $\ce{4H2O}$) on right (balanced)
Wait, let's re - check Option 4 and 5. Wait, maybe I made a mistake. Let's use the correct balancing for the reaction between $\ce{HCl}$ and $\ce{KMnO4}$ (acidic/neutral conditions, but here products are $\ce{Cl2}$, $\ce{MnO2}$, $\ce{H2O}$, $\ce{KCl}$).
The correct balanced reaction is: $\ce{8HCl + 2KMnO4 = 3Cl2 + 2MnO2 + 4H2O + 2KCl}$
Let's verify each atom:
- H: Left: 8 (from $\ce{8HCl}$), Right: \(4\times2 = 8\) (from $\ce{4H2O}$) → Balanced.
- Cl: Left: 8 (from $\ce{8HCl}$), Right: \(3\times2+2 = 8\) (3 $\ce{Cl2}$ molecules have \(3\times2 = 6\) Cl atoms, and 2 $\ce{KCl}$ have 2 Cl atoms; \(6 + 2=8\)) → Balanced.
- Mn: Left: 2 (from $\ce{2KMnO4}$), Right: 2 (from $\ce{2MnO2}$) → Balanced.
- K: Left: 2 (from $\ce{2KMnO4}$), Right: 2 (from $\ce{2KCl}$) → Balanced.
- O: Left: \(2\times4 = 8\) (from $\ce{2KMnO4}$), Right: \(2\times2+4\times1=4 + 4 = 8\) (2 $\ce{MnO2}$ have \(2\times2 = 4\) O atoms, 4 $\ce{H2O}$ have \(4\times1 = 4\) O atoms) → Balanced.
Wait, the fifth option (the last one: $\ce{8HCl + 2KMnO4 -> 3Cl2 + 2MnO2 + 4H2O + 2KCl}$) is balanced. Let's confirm again:
- H: 8 (8 HCl) vs 8 (4 H₂O: 4×2) → Balanced.
- Cl: 8 (8 HCl) vs \(3\times2 + 2=6 + 2 = 8\) (3 Cl₂ and 2 KCl) → Balanced.
- Mn: 2 (2 KMnO₄) vs 2 (2 MnO₂) → Balanced.
- K: 2 (2 KMnO₄) vs 2 (2 KCl) → Balanced.
- O: 8 (2×4 KMnO₄) vs \(2\times2 + 4\times1=4 + 4 = 8\) (2 MnO₂ and…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{8\ce{HCl} + 2\ce{KMnO4}
ightarrow 3\ce{Cl2} + 2\ce{MnO2} + 4\ce{H2O} + 2\ce{KCl}}$ (the last option in the list)