QUESTION IMAGE
Question
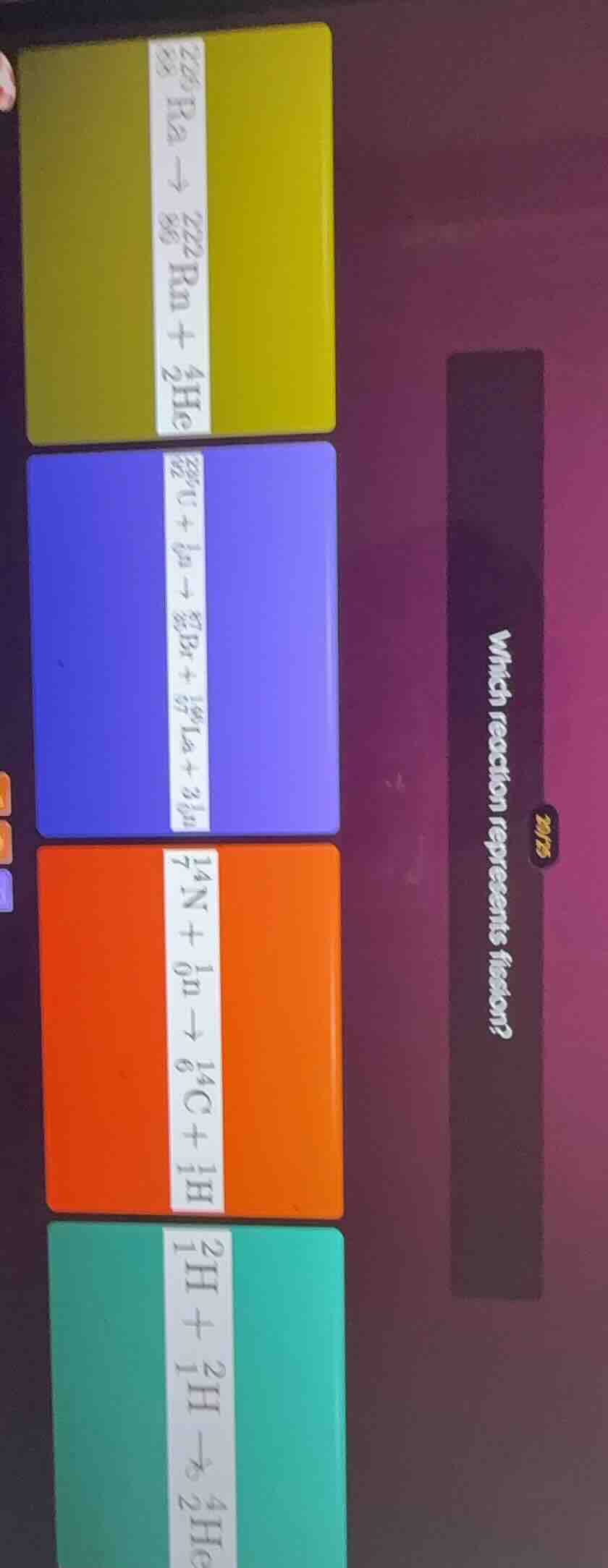
which reaction represents fission?
\ce{_{88}^{226}ra -> _{86}^{222}rn + _{2}^{4}he}
\ce{_{92}^{235}u + _{0}^{1}n -> _{35}^{89}br + _{57}^{144}la + 3_{0}^{1}n}
\ce{_{7}^{14}n + _{0}^{1}n -> _{6}^{14}c + _{1}^{1}h}
\ce{_{1}^{2}h + _{1}^{2}h -> _{2}^{4}he}
Brief Explanations
Nuclear fission is defined as a heavy nucleus splitting into smaller nuclei, often with neutron absorption and release of additional neutrons.
- The yellow card shows alpha decay (a nucleus emits a helium nucleus).
- The blue card shows a heavy uranium nucleus absorbing a neutron, then splitting into smaller bromine and lanthanum nuclei, plus extra neutrons—this matches fission.
- The orange card shows neutron capture and transmutation, not splitting into large fragments.
- The green card shows nuclear fusion (light nuclei combining into a heavier one).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{_{92}^{235}U + _{0}^{1}n
ightarrow _{35}^{87}Br + _{57}^{146}La + 3_{0}^{1}n}$ (the reaction on the blue card)