QUESTION IMAGE
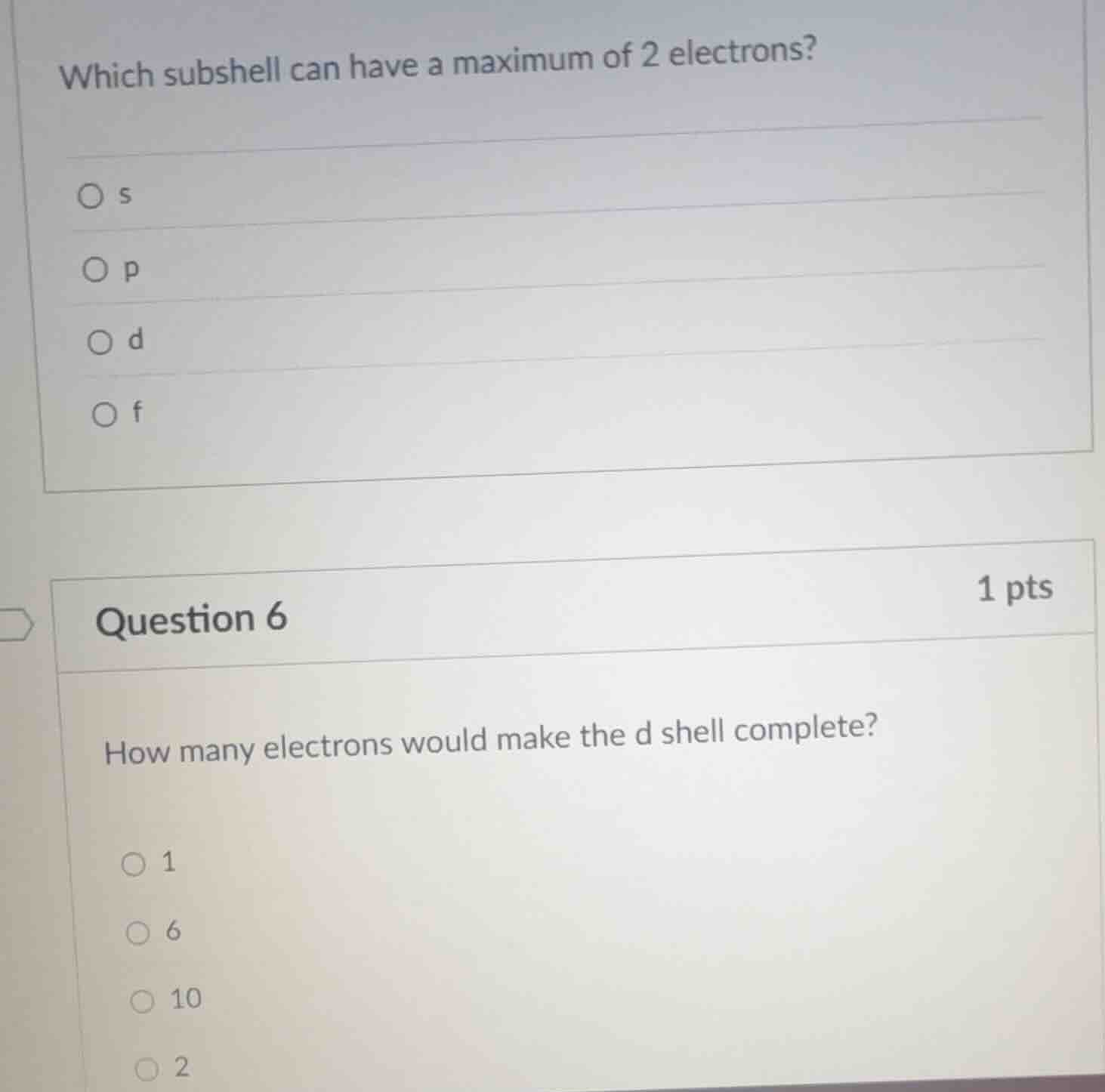
Question
which subshell can have a maximum of 2 electrons?
s
p
d
f
question 6
1 pts
how many electrons would make the d shell complete?
1
6
10
2
First Question (Which subshell can have a maximum of 2 electrons?)
In electron configuration, the s subshell has 1 orbital, and each orbital can hold a maximum of 2 electrons (by Pauli exclusion principle). The p subshell has 3 orbitals (max 6 electrons), d has 5 orbitals (max 10 electrons), and f has 7 orbitals (max 14 electrons). So the s subshell can have a maximum of 2 electrons.
The d subshell has 5 orbitals. Since each orbital can hold a maximum of 2 electrons, the total number of electrons for a complete d subshell is \( 5\times2 = 10 \) electrons.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. s