QUESTION IMAGE
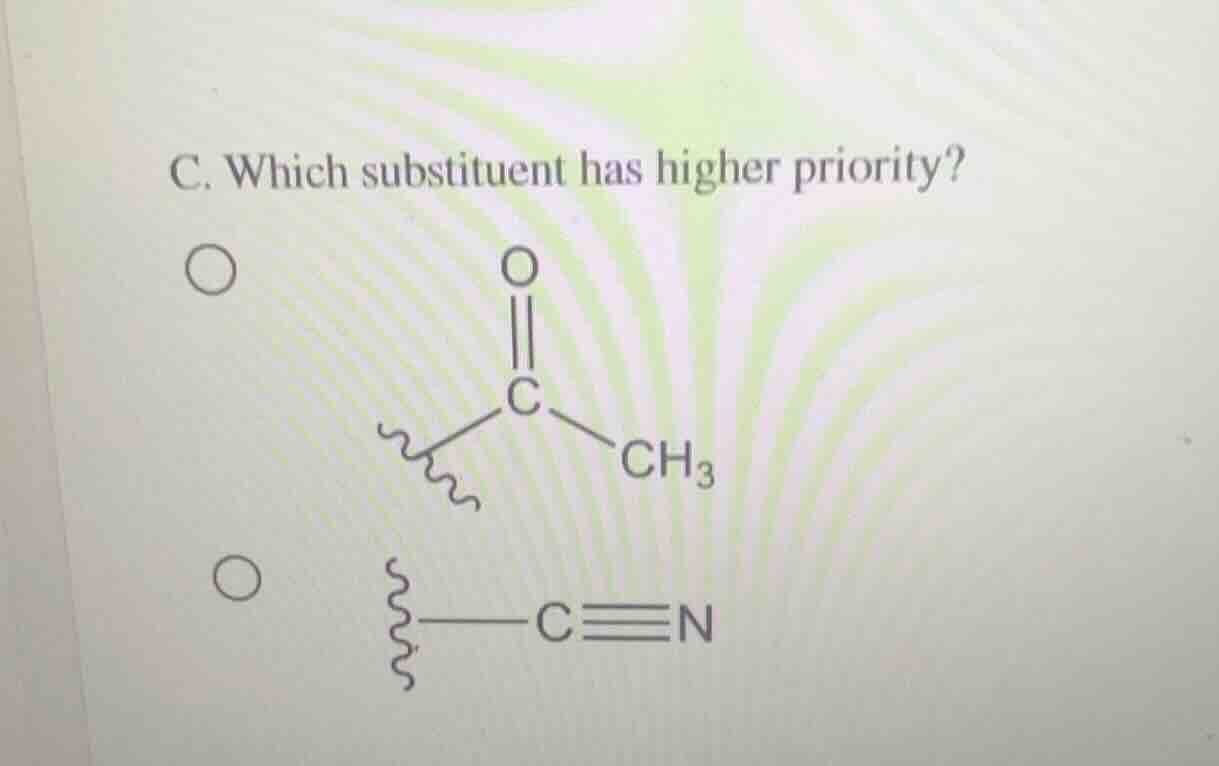
Question
c. which substituent has higher priority?
$ce{(ch3)c(=o)-}$
$ce{-c#n}$
Brief Explanations
Using Cahn-Ingold-Prelog priority rules, we analyze the bonded atoms:
- For the acetyl group ($\text{-C(O)CH}_3$): the central C is double-bonded to O, so it is treated as bonded to 2 O atoms and 1 C (from $\text{CH}_3$).
- For the cyano group ($\text{-C≡N}$): the central C is triple-bonded to N, so it is treated as bonded to 3 N atoms.
O has a higher atomic number (8) than N (7). The double-bonded O in the acetyl group gives it higher priority than the triple-bonded N in the cyano group.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The acetyl group ($\boldsymbol{\text{-C(O)CH}_3}$) has higher priority.