QUESTION IMAGE
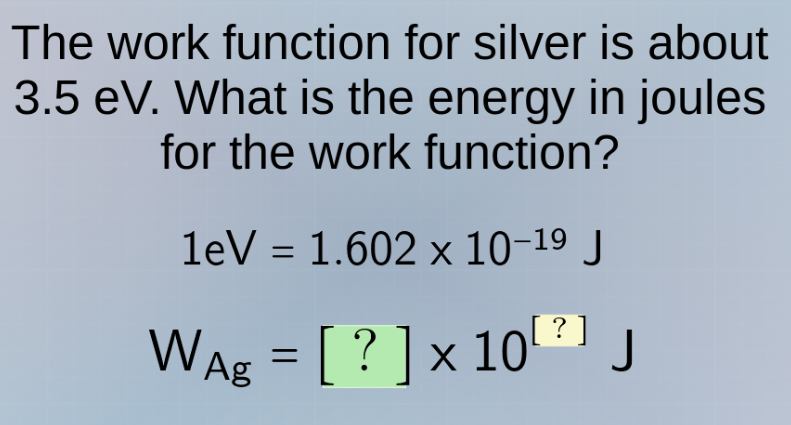
Question
the work function for silver is about 3.5 ev. what is the energy in joules for the work function?
$1\text{ev} = 1.602 \times 10^{-19}\text{ j}$
$w_{\text{ag}} = \square \times 10^{\square}\text{ j}$
Step1: Identify conversion factor
Given $1\ \text{eV} = 1.602 \times 10^{-19}\ \text{J}$
Step2: Convert eV to joules
Multiply work function by conversion factor:
$3.5\ \text{eV} \times 1.602 \times 10^{-19}\ \frac{\text{J}}{\text{eV}}$
Step3: Calculate the product
$3.5 \times 1.602 = 5.607$, so result is $5.607 \times 10^{-19}\ \text{J}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$5.607 \times 10^{-19}\ \text{J}$ (rounded to $5.6 \times 10^{-19}\ \text{J}$ if simplified)