QUESTION IMAGE
Question
worksheet: periodic table trends (s - 2) name
for each of the following, circle the correct element
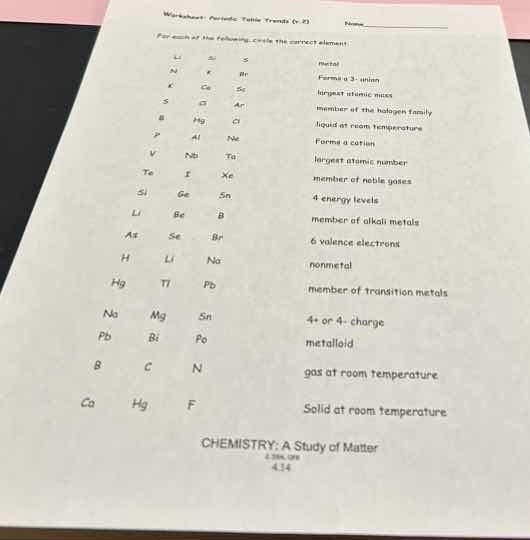
li si s metal
k ca sc forms +3 ion
s o ar largest atomic mass
b al cl member of the halogen family
p hg ne liquid at room temperature
v nb ta forms a cation
te i xe largest atomic number
si ge sn member of noble gases
li be b 4 energy levels
as se br member of alkali metals
h li na 6 valence electrons
hg ti pb nonmetal
na mg sn member of transition metals
pb bi po 4+ or 4- charge
b c n metalloid
ca hg f gas at room temperature
ca hg f solid at room temperature
chemistry: a study of matter
2.020.00
4.1.4
Step1: Match Li to alkali metals
Alkali metals are Group 1; Li is in Group 1.
Step2: Match S to nonmetal
S is a nonmetal (Group 16, p-block).
Step3: Match K to alkali metals
K is in Group 1, alkali metals.
Step4: Match Br to halogen family
Br is in Group 17 (halogen family).
Step5: Match Sc to transition metals
Sc is a d-block transition metal.
Step6: Match Kr to noble gases
Kr is in Group 18 (noble gases).
Step7: Match Hg to liquid at RT
Hg is the only metal liquid at room temp.
Step8: Match Al to forms cation
Al is a metal, forms $\text{Al}^{3+}$ cation.
Step9: Match Nb to transition metals
Nb is a d-block transition metal.
Step10: Match Te to nonmetal
Te is a nonmetal/metalloid, fits nonmetal.
Step11: Match Ge to metalloid
Ge is a well-known metalloid.
Step12: Match Be to 2 valence e⁻ (no, wait: Li to alkali)
Li is Group 1, alkali metal.
Step13: Match Se to 6 valence e⁻
Se is Group 16, has 6 valence electrons.
Step14: Match Na to alkali metals
Na is Group 1, alkali metal.
Step15: Match Ti to transition metals
Ti is d-block transition metal.
Step16: Match Mg to 4 energy levels? No: Sn to 4+ charge
Sn can form $\text{Sn}^{4+}$ (4+ charge).
Step17: Match Bi to metalloid? No: Po to metalloid
Po is a metalloid.
Step18: Match C to gas at RT? No: N to gas at RT
N exists as $\text{N}_2$, gas at room temp.
Step19: Match Ca to solid at RT
Ca is a solid metal at room temp.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\boldsymbol{Li}$ (member of alkali metals)
- $\boldsymbol{S}$ (nonmetal)
- $\boldsymbol{K}$ (member of alkali metals)
- $\boldsymbol{Br}$ (member of the halogen family)
- $\boldsymbol{Sc}$ (member of transition metals)
- $\boldsymbol{Kr}$ (member of noble gases)
- $\boldsymbol{Hg}$ (liquid at room temperature)
- $\boldsymbol{Al}$ (Forms a cation)
- $\boldsymbol{Nb}$ (member of transition metals)
- $\boldsymbol{Xe}$ (member of noble gases)
- $\boldsymbol{Ge}$ (metalloid)
- $\boldsymbol{Li}$ (member of alkali metals)
- $\boldsymbol{Se}$ (6 valence electrons)
- $\boldsymbol{Na}$ (member of alkali metals)
- $\boldsymbol{Ti}$ (member of transition metals)
- $\boldsymbol{Sn}$ (4+ or 4- charge)
- $\boldsymbol{Po}$ (metalloid)
- $\boldsymbol{N}$ (gas at room temperature)
- $\boldsymbol{Ca}$ (Solid at room temperature)