QUESTION IMAGE
Question
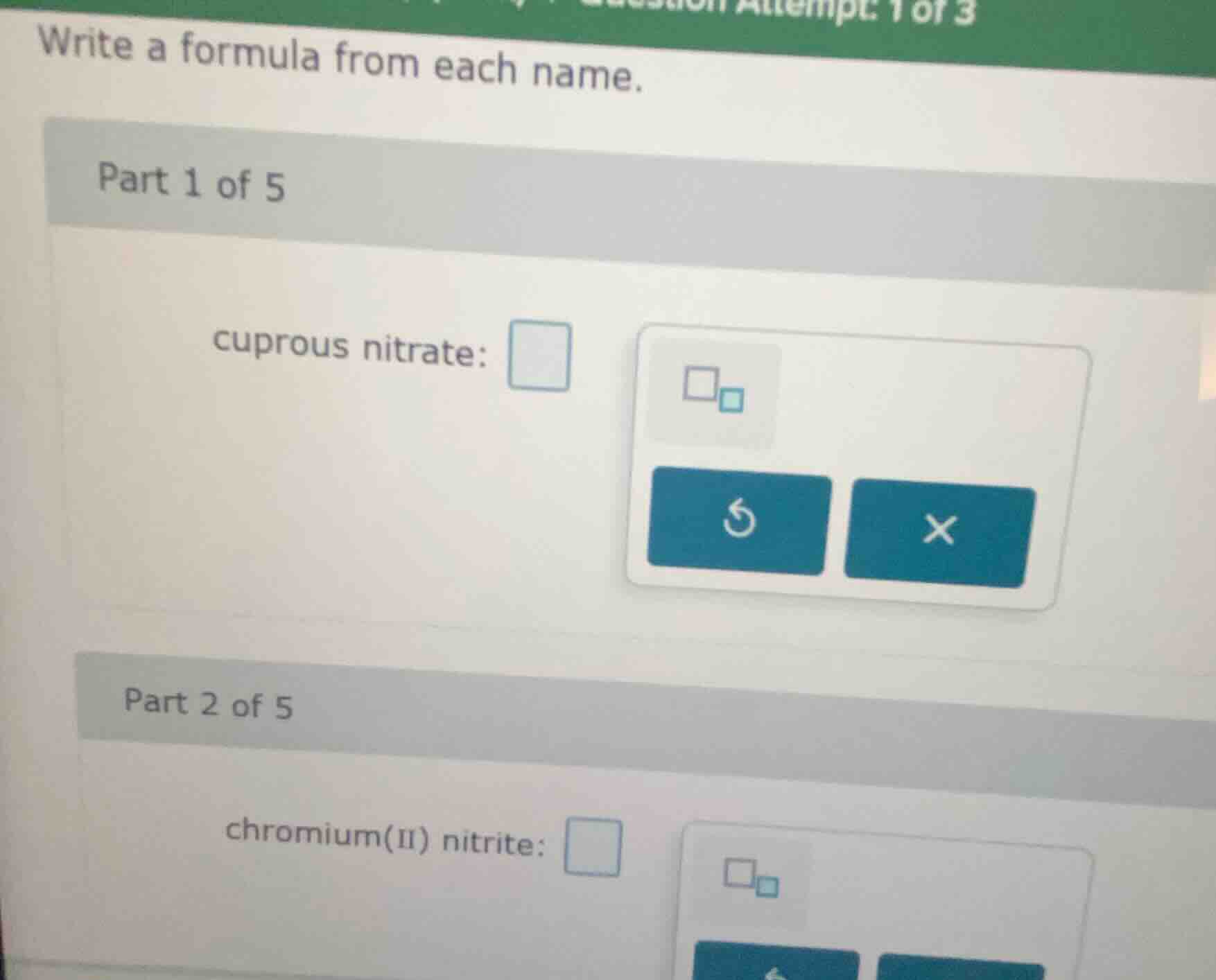
write a formula from each name.
part 1 of 5
cuprous nitrate:
part 2 of 5
chromium(ii) nitrite:
Response
Part 1 of 5: Cuprous Nitrate
Step 1: Identify ions
Cuprous is $Cu^+$ (copper(I) ion), nitrate is $NO_3^-$.
Step 2: Balance charges
To balance $+1$ (from $Cu^+$) and $-1$ (from $NO_3^-$), we need 1 $Cu^+$ and 1 $NO_3^-$.
Step 1: Identify ions
Chromium(II) is $Cr^{2+}$, nitrite is $NO_2^-$.
Step 2: Balance charges
The charge of $Cr^{2+}$ is $+2$, and $NO_2^-$ is $-1$. To balance, we need 1 $Cr^{2+}$ and 2 $NO_2^-$ (since $2\times(-1) = -2$ to balance $+2$).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{CuNO_3}$