QUESTION IMAGE
Question
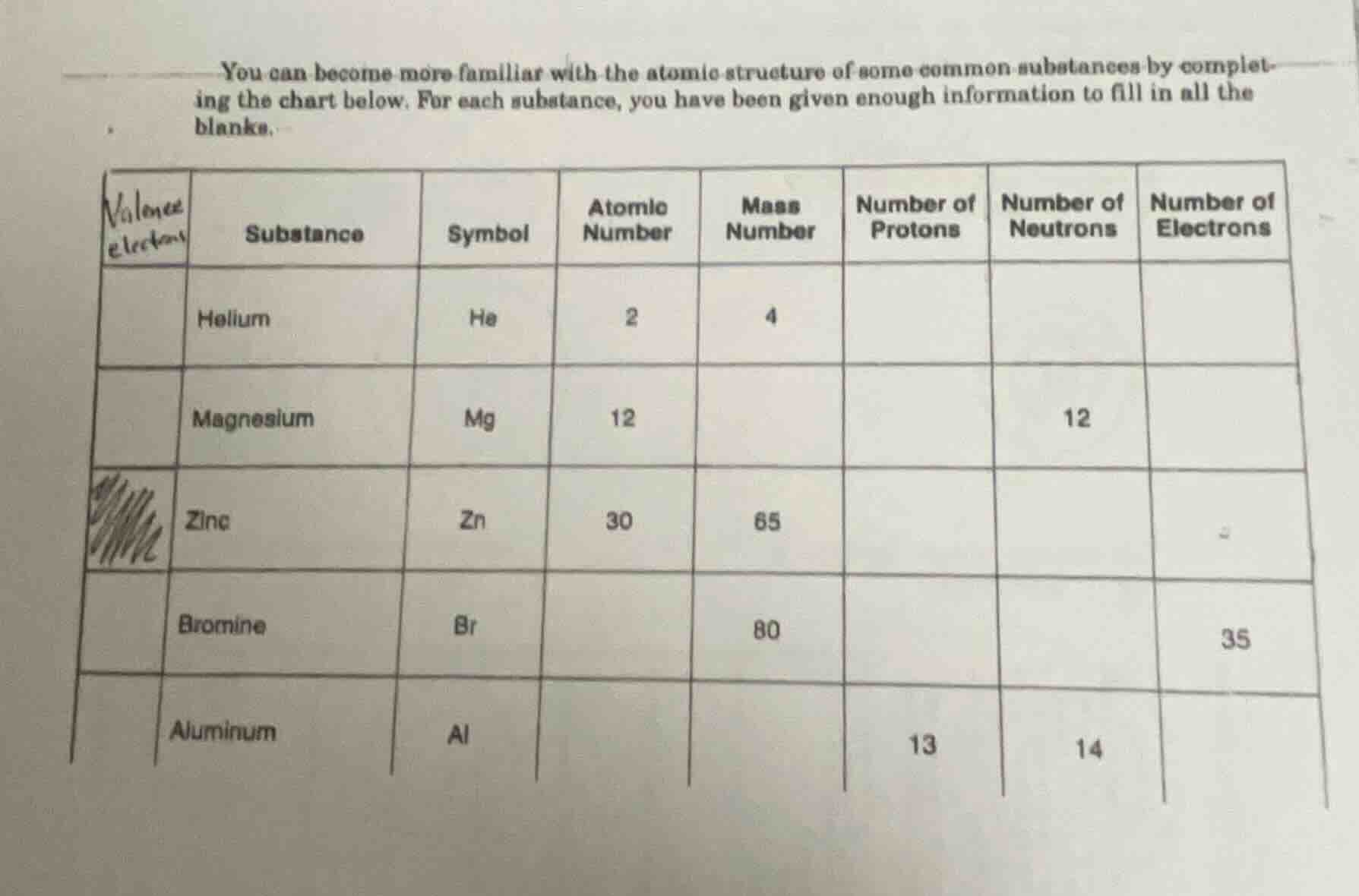
you can become more familiar with the atomic structure of some common substances by completing the chart below. for each substance, you have been given enough information to fill in all the blanks.
substance | symbol | atomic number | mass number | number of protons | number of neutrons | number of electrons
helium | he | 2 | 4 | | |
magnesium | mg | 12 | | | 12 |
zinc | zn | 30 | 65 | | |
bromine | br | | 80 | | | 35
aluminum | al | | | 13 | 14 |
Step1: Recall atomic rules
Key relationships:
- Atomic Number = Number of Protons = Number of Electrons (neutral atom)
- Mass Number = Protons + Neutrons
- Valence electrons: Group number (main group elements)
Step2: Fill Helium row
Protons = Atomic Number = $2$
Neutrons = $4-2=2$
Electrons = Protons = $2$
Valence electrons = $2$ (full outer shell)
Step3: Fill Magnesium row
Protons = Atomic Number = $12$
Mass Number = $12+12=24$
Electrons = Protons = $12$
Valence electrons = $2$ (Group 2)
Step4: Fill Zinc row
Protons = Atomic Number = $30$
Neutrons = $65-30=35$
Electrons = Protons = $30$
Valence electrons = $2$ (Group 12)
Step5: Fill Bromine row
Atomic Number = Electrons = $35$
Protons = Atomic Number = $35$
Neutrons = $80-35=45$
Valence electrons = $7$ (Group 17)
Step6: Fill Aluminum row
Atomic Number = Protons = $13$
Mass Number = $13+14=27$
Electrons = Protons = $13$
Valence electrons = $3$ (Group 13)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Valence Electrons | Substance | Symbol | Atomic Number | Mass Number | Number of Protons | Number of Neutrons | Number of Electrons |
|---|---|---|---|---|---|---|---|
| 2 | Magnesium | Mg | 12 | 24 | 12 | 12 | 12 |
| 2 | Zinc | Zn | 30 | 65 | 30 | 35 | 30 |
| 7 | Bromine | Br | 35 | 80 | 35 | 45 | 35 |
| 3 | Aluminum | Al | 13 | 27 | 13 | 14 | 13 |