QUESTION IMAGE
Question
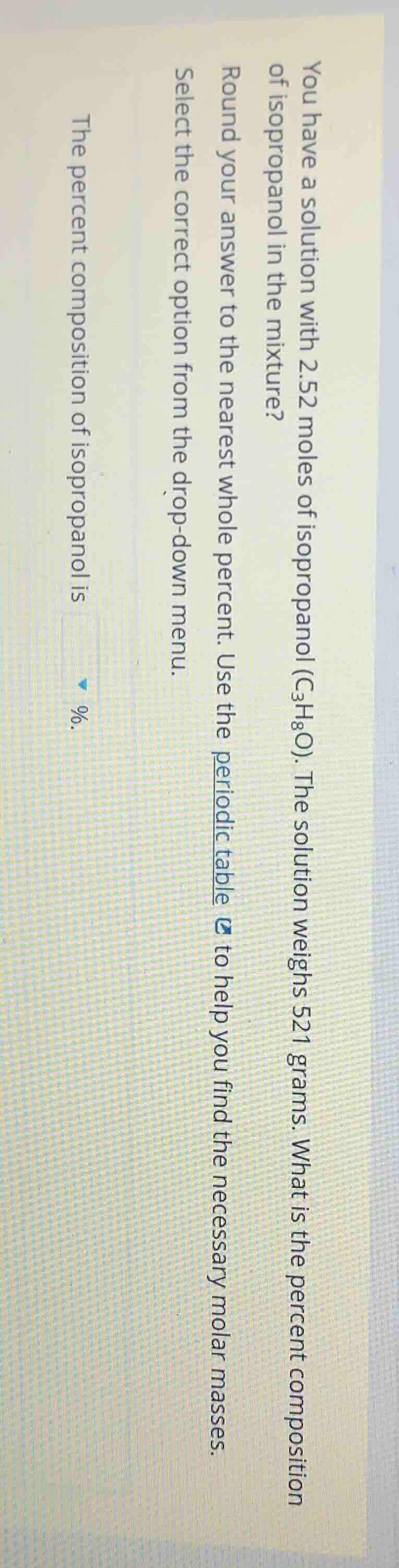
you have a solution with 2.52 moles of isopropanol (c₃h₈o). the solution weighs 521 grams. what is the percent composition of isopropanol in the mixture? round your answer to the nearest whole percent. use the periodic table to help you find the necessary molar masses. select the correct option from the drop - down menu. the percent composition of isopropanol is %.
Step1: Calculate molar mass of isopropanol ($\ce{C3H8O}$)
Molar mass of C = 12.01 g/mol, H = 1.008 g/mol, O = 16.00 g/mol.
Molar mass = \( 3\times12.01 + 8\times1.008 + 16.00 \)
= \( 36.03 + 8.064 + 16.00 \)
= \( 60.094 \) g/mol.
Step2: Calculate mass of isopropanol
Moles of isopropanol = 2.52 mol.
Mass = moles × molar mass = \( 2.52 \times 60.094 \)
≈ \( 151.43688 \) g.
Step3: Calculate percent composition
Percent composition = \( \frac{\text{Mass of isopropanol}}{\text{Mass of solution}} \times 100 \)
= \( \frac{151.43688}{521} \times 100 \)
≈ \( 29.0666\% \), rounded to nearest whole percent is 29%.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
29%