QUESTION IMAGE
Question
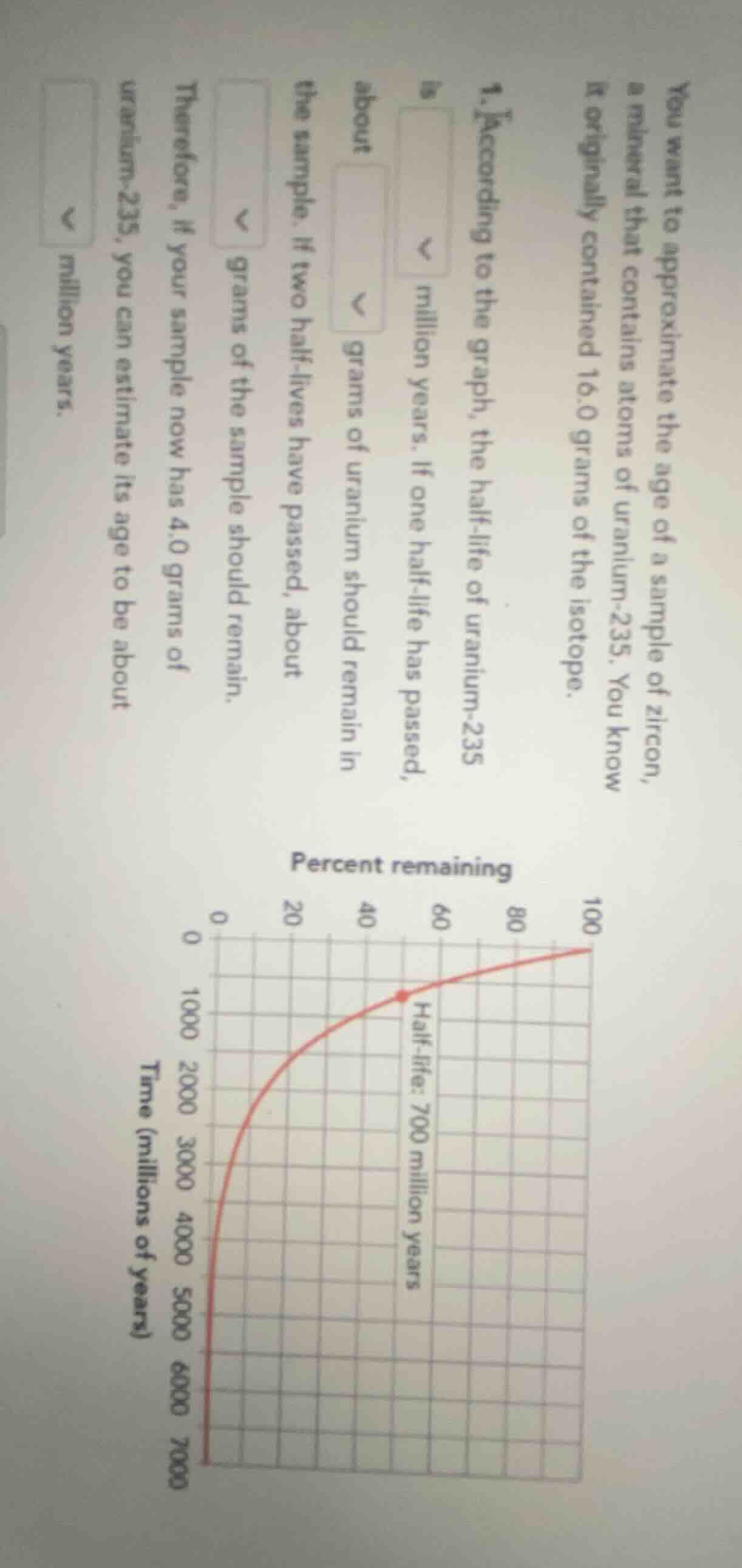
you want to approximate the age of a sample of zircon, a mineral that contains atoms of uranium-235. you know it originally contained 16.0 grams of the isotope.
- according to the graph, the half-life of uranium-235 is \\(\boldsymbol{\downarrow}\\) million years. if one half-life has passed, about \\(\boldsymbol{\downarrow}\\) grams of uranium should remain in the sample. if two half-lives have passed, about \\(\boldsymbol{\downarrow}\\) grams of the sample should remain.
therefore, if your sample now has 4.0 grams of uranium-235, you can estimate its age to be about \\(\boldsymbol{\downarrow}\\) million years.
graph: percent remaining (y-axis: 0 - 100) vs time (millions of years, x-axis: 0 - 7000). curve labeled half - life: 700 million years
Step1: Identify half - life
From the graph, the half - life of uranium - 235 is 700 million years.
Step2: Calculate remaining after 1 half - life
The original amount \(N_0 = 16.0\) grams. After one half - life, the remaining amount \(N\) is given by the formula \(N=\frac{N_0}{2}\). So \(N=\frac{16.0}{2}=8.0\) grams.
Step3: Calculate remaining after 2 half - lives
After two half - lives, the remaining amount \(N\) is given by \(N = \frac{N_0}{2^2}\). Substituting \(N_0 = 16.0\) grams, we get \(N=\frac{16.0}{4}=4.0\) grams.
Step4: Calculate age for 4.0 grams remaining
We know that the remaining amount \(N = 4.0\) grams and \(N_0=16.0\) grams. Let the number of half - lives be \(n\). We have \(N = N_0\times(\frac{1}{2})^n\). Substituting the values, \(4.0=16.0\times(\frac{1}{2})^n\). Dividing both sides by 16.0, we get \(\frac{4.0}{16.0}=(\frac{1}{2})^n\), or \(\frac{1}{4}=(\frac{1}{2})^n\). Since \(\frac{1}{4}=(\frac{1}{2})^2\), \(n = 2\). The age of the sample is \(n\times\) half - life. So age \(=2\times700 = 1400\) million years.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- The half - life of uranium - 235 is 700 million years.
- After one half - life, about 8.0 grams of uranium should remain.
- After two half - lives, about 4.0 grams of the sample should remain.
- If the sample now has 4.0 grams of uranium - 235, its age is about 1400 million years.