QUESTION IMAGE
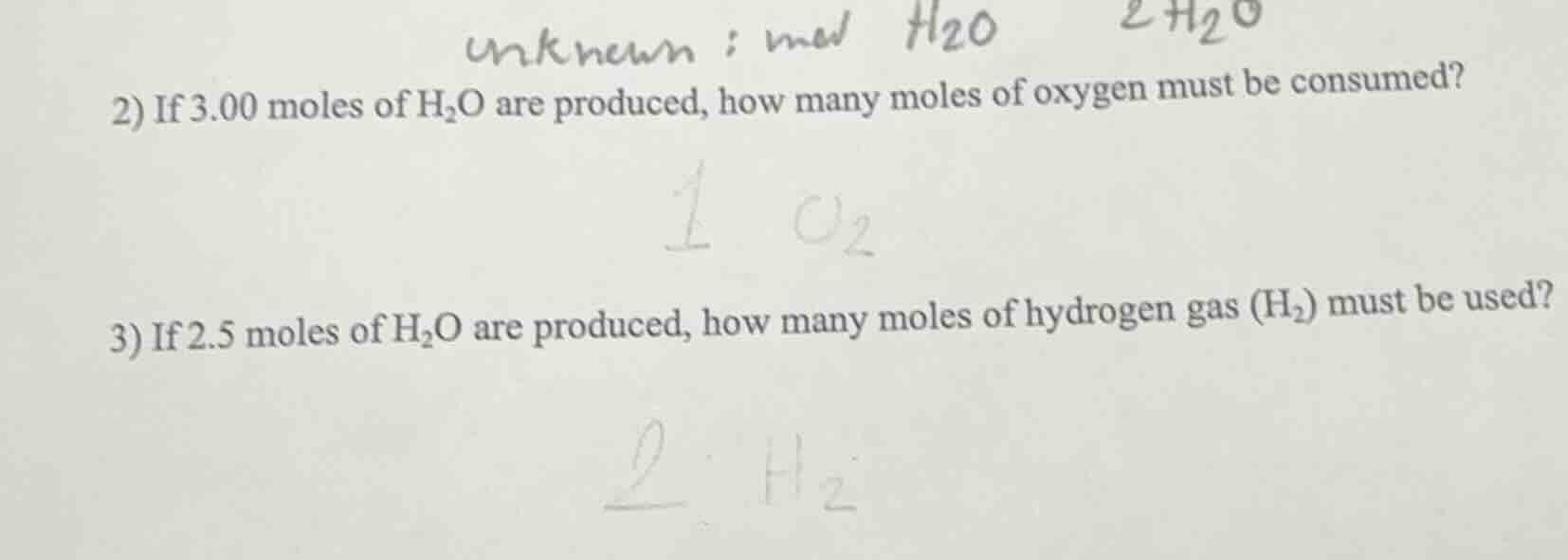
Question
- if 3.00 moles of h₂o are produced, how many moles of oxygen must be consumed?
- if 2.5 moles of h₂o are produced, how many moles of hydrogen gas (h₂) must be used?
Step1: Balanced reaction for water formation
The synthesis of water follows:
$$2\text{H}_2 + \text{O}_2
ightarrow 2\text{H}_2\text{O}$$
---
For Question 2:
Step2: Mole ratio of $\text{O}_2$ to $\text{H}_2\text{O}$
From the reaction, $\frac{n(\text{O}_2)}{n(\text{H}_2\text{O})} = \frac{1}{2}$
Step3: Calculate moles of $\text{O}_2$
$$n(\text{O}_2) = \frac{1}{2} \times n(\text{H}_2\text{O}) = \frac{1}{2} \times 3.00 = 1.50$$
---
For Question 3:
Step2: Mole ratio of $\text{H}_2$ to $\text{H}_2\text{O}$
From the reaction, $\frac{n(\text{H}_2)}{n(\text{H}_2\text{O})} = \frac{2}{2} = 1$
Step3: Calculate moles of $\text{H}_2$
$$n(\text{H}_2) = 1 \times n(\text{H}_2\text{O}) = 1 \times 2.5 = 2.5$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- 1.50 moles of $\text{O}_2$
- 2.5 moles of $\text{H}_2$