QUESTION IMAGE
Question
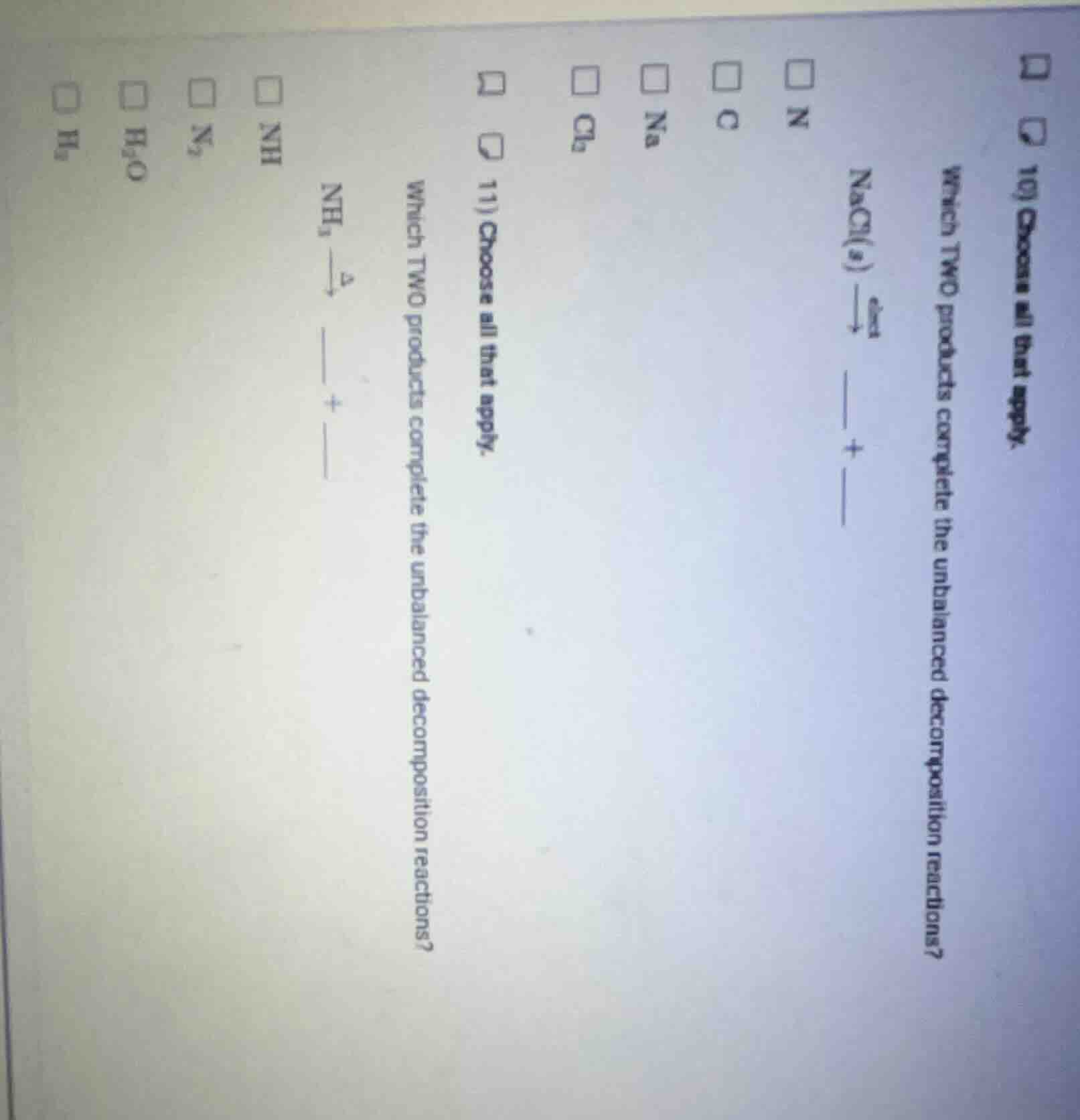
- choose all that apply. which two products complete the unbalanced decomposition reactions? nacl(s) $stackrel{elect}{
ightarrow}$ ___ + ___ $square$ n $square$ c $square$ na $square$ cl₂ 11) choose all that apply. which two products complete the unbalanced decomposition reactions? nh₃ $stackrel{delta}{
ightarrow}$ ___ + ___ $square$ nh $square$ n₂ $square$ h₂o $square$ h₂
Step1: Analyze NaCl decomposition
Electrolysis of solid NaCl breaks into its elements:
$$\text{NaCl}(s) \xrightarrow{\text{elect.}} \text{Na} + \text{Cl}_2$$
Step2: Analyze NH₃ decomposition
Heated ammonia decomposes into nitrogen and hydrogen:
$$\text{NH}_3 \xrightarrow{\Delta} \text{N}_2 + \text{H}_2$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
For question 10:
□ N
□ C
☑ Na
☑ Cl₂
For question 11:
□ NH
☑ N₂
□ H₂O
☑ H₂