QUESTION IMAGE
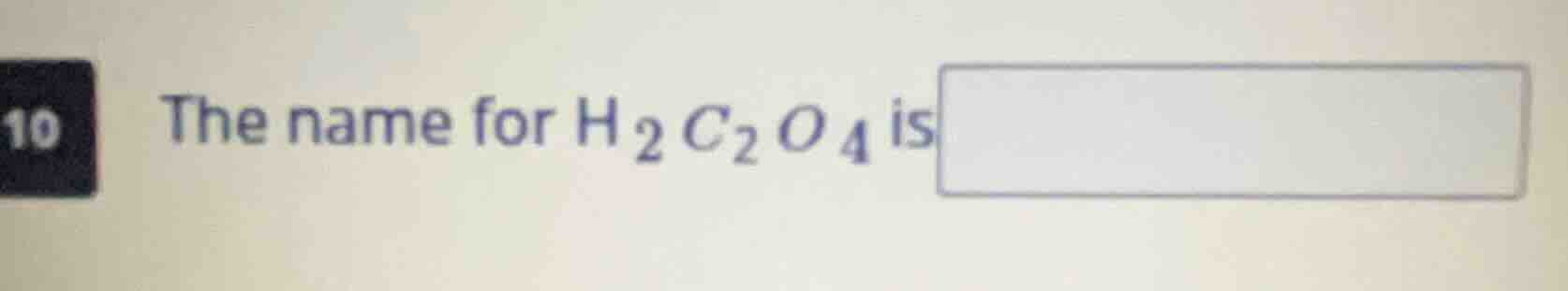
Question
10 the name for $h_2c_2o_4$ is
Step1: Identify the compound type
$H_2C_2O_4$ is an acid. The anion here is $C_2O_4^{2 - }$, which is the oxalate ion. When an acid has the hydrogen - oxalate anion, or in the case of the fully protonated form, we name it based on the anion. The oxalate ion comes from oxalic acid.
Step2: Name the acid
For an acid formed from the oxalate ion ($C_2O_4^{2 - }$), the formula $H_2C_2O_4$ represents oxalic acid (also known as ethanedioic acid, but the common name is oxalic acid).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
oxalic acid (or ethanedioic acid)