QUESTION IMAGE
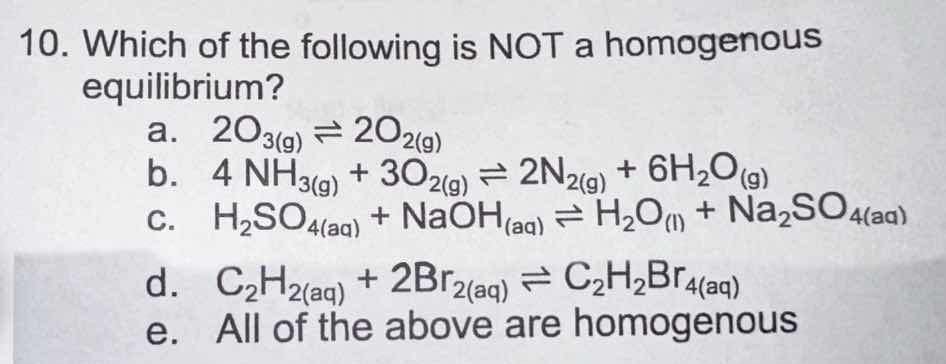
Question
- which of the following is not a homogenous equilibrium?
a. \\( 2o_3(g) \
ightleftharpoons 2o_2(g) \\)
b. \\( 4 nh_3(g) + 3o_2(g) \
ightleftharpoons 2n_2(g) + 6h_2o_{(g)} \\)
c. \\( h_2so_4(aq) + naoh_{(aq)} \
ightleftharpoons h_2o_{(l)} + na_2so_4(aq) \\)
d. \\( c_2h_2(aq) + 2br_2(aq) \
ightleftharpoons c_2h_2br_4(aq) \\)
e. all of the above are homogenous
Step1: Recall Homogeneous Equilibrium
A homogeneous equilibrium is one where all reactants and products are in the same phase (gas, liquid, aqueous, solid).
Step2: Analyze Option a
In \( 2O_3(g)
ightleftharpoons 2O_2(g) \), both reactant and product are in the gas phase. So, it is homogeneous.
Step3: Analyze Option b
For \( 4NH_3(g) + 3O_2(g)
ightleftharpoons 2N_2(g) + 6H_2O(g) \), all species are in the gas phase. Thus, homogeneous.
Step4: Analyze Option c
In \( H_2SO_4(aq) + NaOH(aq)
ightleftharpoons H_2O(l) + Na_2SO_4(aq) \), the reactants are aqueous, but one product (\( H_2O \)) is in the liquid phase and others are aqueous. Since phases are not all the same (aqueous vs liquid), this is not homogeneous. Wait, but wait, actually, in aqueous solutions, water is often considered as part of the solvent, but strictly, the phases: \( H_2SO_4 \) (aq), \( NaOH \) (aq) are aqueous (dissolved in water), \( H_2O \) (l) is liquid, \( Na_2SO_4 \) (aq) is aqueous. So phases are mixed (aqueous and liquid). So this is not homogeneous. But wait, let's check option d.
Step5: Analyze Option d
In \( C_2H_2(aq) + 2Br_2(aq)
ightleftharpoons C_2H_2Br_4(aq) \), all species are in the aqueous phase. So homogeneous.
Wait, but earlier analysis of option c: Wait, maybe I made a mistake. Wait, \( H_2O(l) \) in the reaction: in aqueous reactions, sometimes water is considered as a solvent, but the standard definition is about phases of reactants and products. Let's re - check:
Homogeneous equilibrium: all reactants and products in the same phase.
Option a: all gas - same phase.
Option b: all gas - same phase.
Option c: reactants are aqueous (dissolved in water), products: \( H_2O \) is liquid, \( Na_2SO_4 \) is aqueous. So phases: aqueous (reactants), liquid and aqueous (products). So not all same phase.
Option d: all aqueous - same phase.
So option c is not a homogeneous equilibrium? But wait, let's check again. Wait, the reaction is \( H_2SO_4(aq)+NaOH(aq)
ightleftharpoons H_2O(l)+Na_2SO_4(aq) \). The reactants are in aqueous phase (dissolved in water), one product is in liquid phase and the other in aqueous. So the phases of reactants and products are not all the same. So option c is not homogeneous. But wait, maybe the question has a different take. Wait, maybe the answer is c? But let's check the options again.
Wait, maybe I messed up. Let's re - evaluate:
Wait, homogeneous equilibrium: all substances (reactants and products) in the same phase.
Option a: gas - gas: same.
Option b: gas - gas: same.
Option c: aqueous (reactants) and (liquid + aqueous) products. So different phases.
Option d: aqueous - aqueous: same.
So the one that is NOT homogeneous is option c? But wait, the original options: let's check the problem again.
Wait, the problem is asking which is NOT a homogeneous equilibrium.
So the answer should be c? But wait, let's check the options again. Wait, maybe I made a mistake in option c. Wait, \( H_2O(l) \) and the aqueous solutions: in the reaction, \( H_2SO_4 \) and \( NaOH \) are dissolved in water (aqueous), and the products are \( H_2O \) (liquid) and \( Na_2SO_4 \) dissolved in water (aqueous). So the phases are aqueous (reactants and \( Na_2SO_4 \)) and liquid (\( H_2O \)). So they are not all in the same phase. So option c is not a homogeneous equilibrium. But wait, let's check the other options again.
Wait, option d: all aqueous, so homogeneous.
Option a: all gas, homogeneous.
Option b: all gas, homogeneous.
So the answer is c? Wait, but let's check the options again. The options are a to e. So t…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
c. \( \ce{H_2SO_4(aq) + NaOH(aq)
ightleftharpoons H_2O(l) + Na_2SO_4(aq)} \)