QUESTION IMAGE
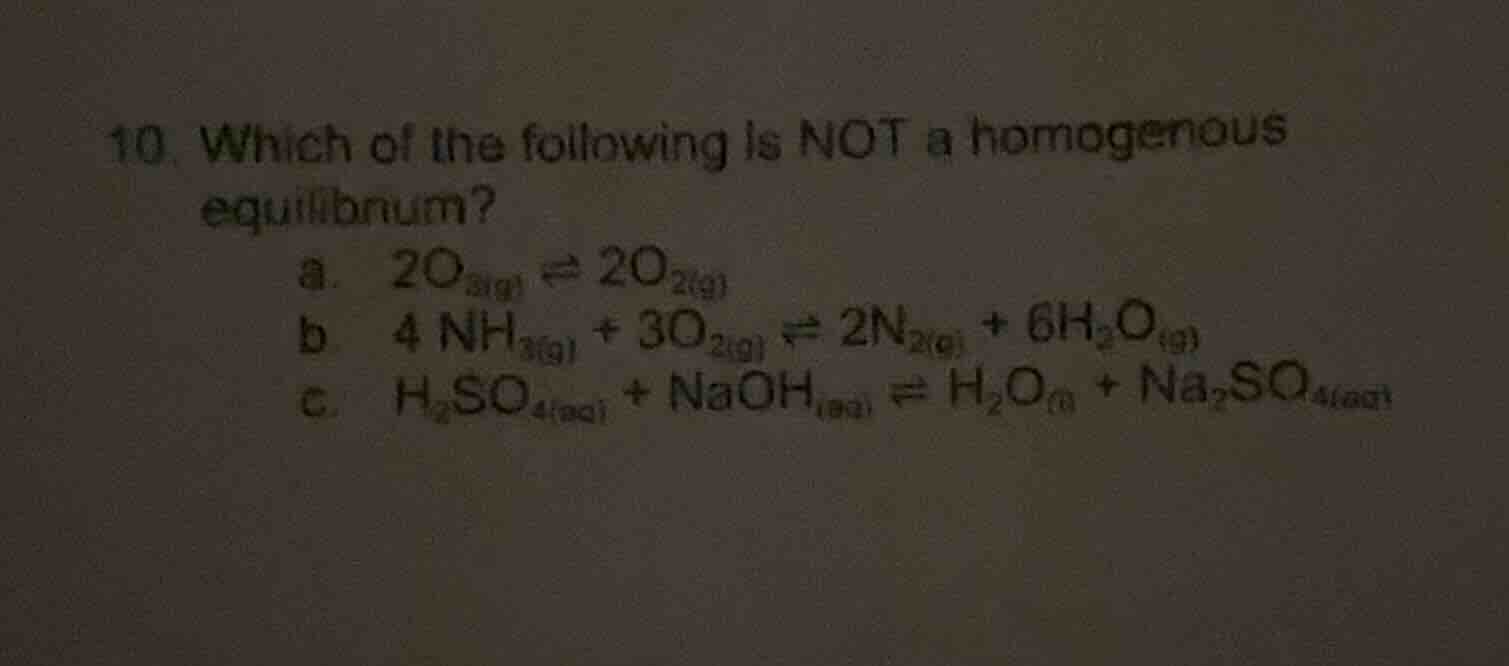
Question
- which of the following is not a homogenous equilibrium?
a. $2o_{3(g)} \
ightleftharpoons 2o_{2(g)}$
b. $4nh_{3(g)} + 3o_{2(g)} \
ightleftharpoons 2n_{2(g)} + 6h_2o_{(g)}$
c. $h_2so_{4(aq)} + naoh_{(aq)} \
ightleftharpoons h_2o_{(l)} + na_2so_{4(aq)}$
A homogeneous equilibrium has all reactants and products in the same phase. In option a, all species are gaseous ($\ce{O3(g)}$ and $\ce{O2(g)}$). In option b, all species are gaseous ($\ce{NH3(g)}$, $\ce{O2(g)}$, $\ce{N2(g)}$, $\ce{H2O(g)}$). In option c, $\ce{H2O}$ is in the liquid phase while others are aqueous ($\ce{H2SO4(aq)}$, $\ce{NaOH(aq)}$, $\ce{Na2SO4(aq)}$), so it is not homogeneous.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
c. $\ce{H_{2}SO_{4(aq)} + NaOH_{(aq)}
ightleftharpoons H_{2}O_{(l)} + Na_{2}SO_{4(aq)}}$