QUESTION IMAGE
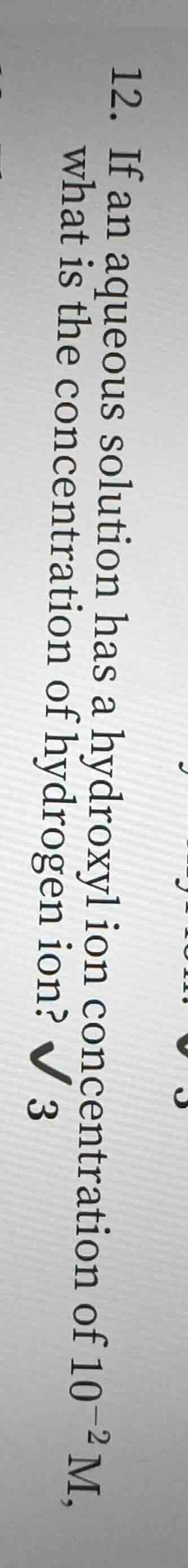
Question
- if an aqueous solution has a hydroxyl ion concentration of $10^{-2}$ m, what is the concentration of hydrogen ion? $\boldsymbol{sqrt{3}}$
Step1: Use water autoionization constant
At 25°C, $K_w = [\text{H}^+][\text{OH}^-] = 1.0\times10^{-14}$
Step2: Isolate $[\text{H}^+]$ and substitute values
Rearrange to solve for hydrogen ion concentration: $[\text{H}^+] = \frac{K_w}{[\text{OH}^-]}$
Substitute $K_w=1.0\times10^{-14}$ and $[\text{OH}^-]=10^{-2}\ \text{M}$:
$[\text{H}^+] = \frac{1.0\times10^{-14}}{10^{-2}}$
Step3: Calculate the final value
Simplify the expression: $[\text{H}^+] = 1.0\times10^{-12}\ \text{M}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$1.0\times10^{-12}\ \text{M}$