QUESTION IMAGE
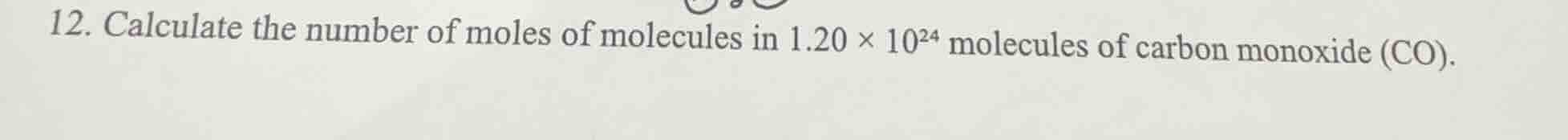
Question
- calculate the number of moles of molecules in $1.20 \times 10^{24}$ molecules of carbon monoxide (co).
Step1: Recall Avogadro's constant
Avogadro's number $N_A = 6.02 \times 10^{23}$ molecules/mol.
Step2: Use mole-molecule conversion formula
Moles $n = \frac{\text{Number of molecules}}{N_A}$
$$n = \frac{1.20 \times 10^{24}}{6.02 \times 10^{23}}$$
Step3: Calculate the result
$$n \approx 1.99 \approx 2.00$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The number of moles is approximately $\boldsymbol{2.00}$ mol (or $\boldsymbol{1.99}$ mol for more precision).