QUESTION IMAGE
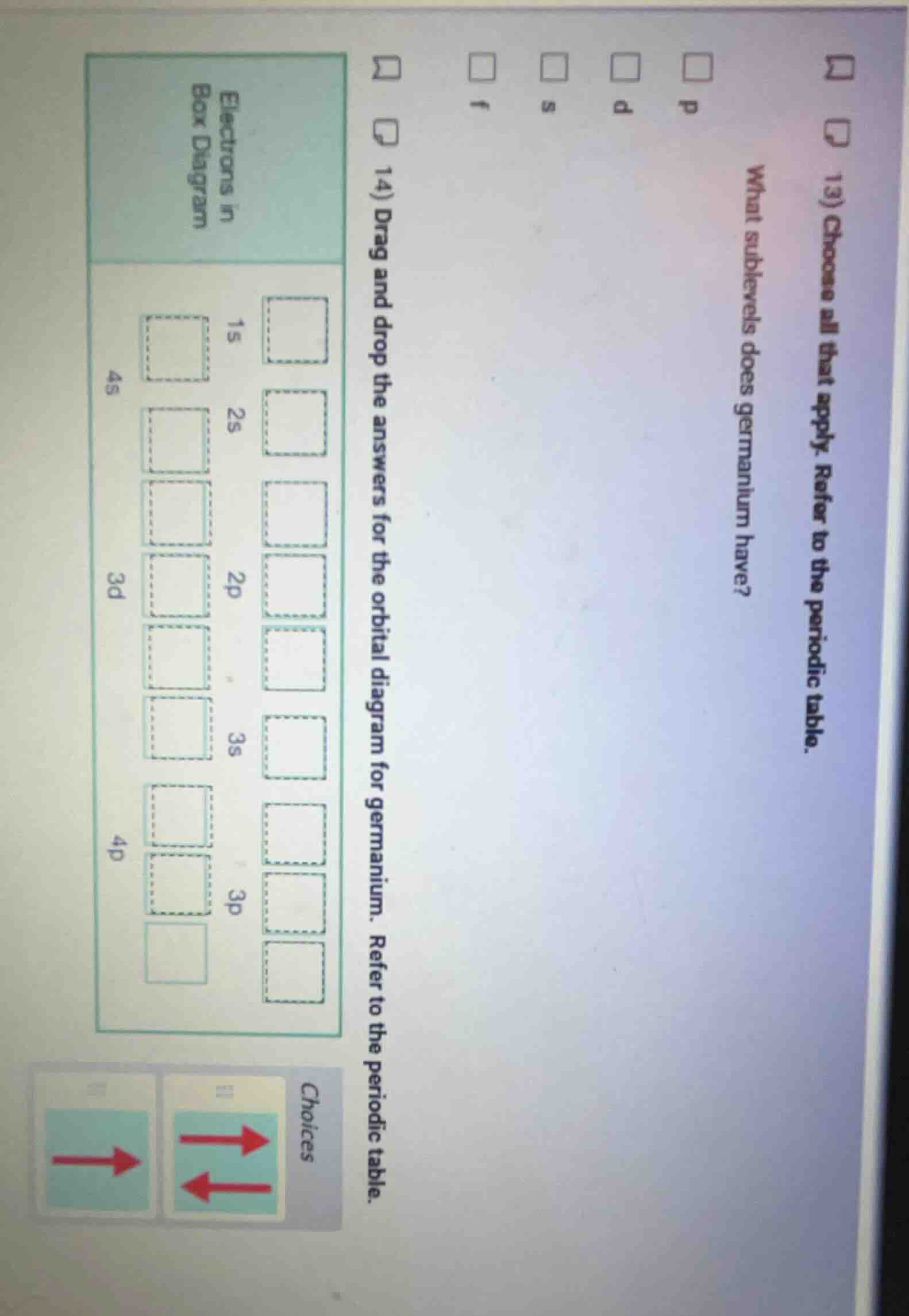
Question
- choose all that apply. refer to the periodic table.
what sublevels does germanium have?
p d s f
- drag and drop the answers for the orbital diagram for germanium. refer to the periodic table.
electrons in
box diagram
1s 2s 2p 3s 3p
4s 3d 4p
choices
Step1: Find Ge's atomic number
Germanium (Ge) has an atomic number of 32, so it has 32 electrons.
Step2: List occupied sublevels
Using the Aufbau principle, the electron configuration of Ge is $1s^22s^22p^63s^23p^64s^23d^{10}4p^2$. The sublevels here are s, p, d.
Step3: Fill orbital diagram (Hund's rule)
- 1s: 2 paired electrons ($\uparrow\downarrow$)
- 2s: 2 paired electrons ($\uparrow\downarrow$)
- 2p: 6 electrons (3 orbitals, each with $\uparrow\downarrow$)
- 3s: 2 paired electrons ($\uparrow\downarrow$)
- 3p: 6 electrons (3 orbitals, each with $\uparrow\downarrow$)
- 4s: 2 paired electrons ($\uparrow\downarrow$)
- 3d: 10 electrons (5 orbitals, each with $\uparrow\downarrow$)
- 4p: 2 unpaired electrons (2 orbitals, each with $\uparrow$)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
13)
- $\square$ p
- $\square$ d
- $\square$ s
- $\square$ f (unselected)
- Orbital diagram filled as:
- 1s: $\boldsymbol{\uparrow\downarrow}$
- 2s: $\boldsymbol{\uparrow\downarrow}$
- 2p: $\boldsymbol{\uparrow\downarrow}$, $\boldsymbol{\uparrow\downarrow}$, $\boldsymbol{\uparrow\downarrow}$
- 3s: $\boldsymbol{\uparrow\downarrow}$
- 3p: $\boldsymbol{\uparrow\downarrow}$, $\boldsymbol{\uparrow\downarrow}$, $\boldsymbol{\uparrow\downarrow}$
- 4s: $\boldsymbol{\uparrow\downarrow}$
- 3d: $\boldsymbol{\uparrow\downarrow}$, $\boldsymbol{\uparrow\downarrow}$, $\boldsymbol{\uparrow\downarrow}$, $\boldsymbol{\uparrow\downarrow}$, $\boldsymbol{\uparrow\downarrow}$
- 4p: $\boldsymbol{\uparrow}$, $\boldsymbol{\uparrow}$, $\boldsymbol{\square}$ (empty)