QUESTION IMAGE
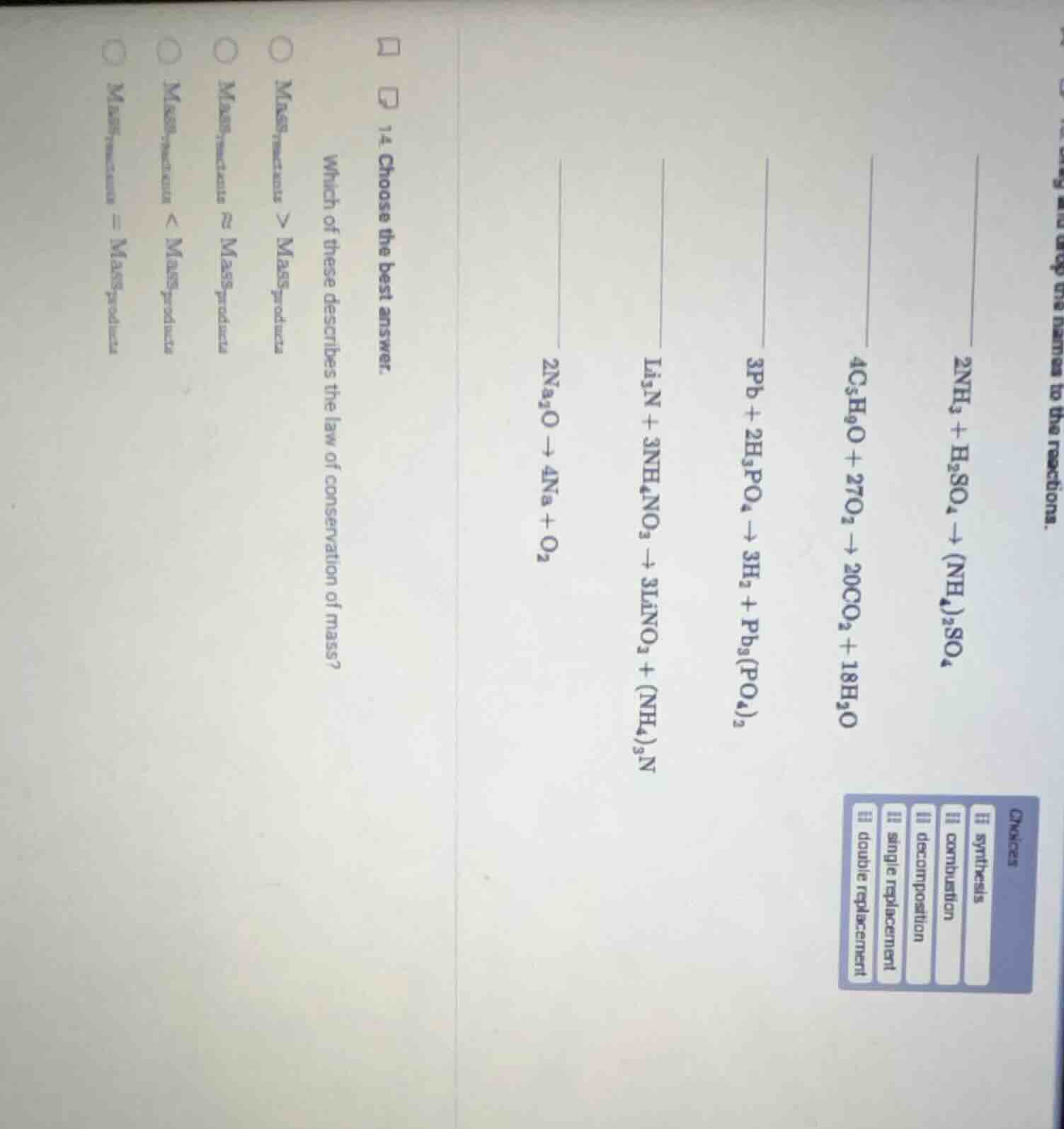
Question
- choose the best answer. which of these describes the law of conservation of mass? mass$_{reactants}$ > mass$_{products}$ mass$_{reactants}$ ≈ mass$_{products}$ mass$_{reactants}$ < mass$_{products}$ mass$_{reactants}$ = mass$_{products}$
(for reaction classification):
Step1: Identify reaction 1 type
$2\text{NH}_3 + \text{H}_2\text{SO}_4
ightarrow (\text{NH}_4)_2\text{SO}_4$: Two reactants form one product, so it is synthesis.
Step2: Identify reaction 2 type
$4\text{C}_5\text{H}_9\text{O} + 27\text{O}_2
ightarrow 20\text{CO}_2 + 18\text{H}_2\text{O}$: A compound reacts with $\text{O}_2$ to produce $\text{CO}_2$ and $\text{H}_2\text{O}$, so it is combustion.
Step3: Identify reaction 3 type
$3\text{Pb} + 2\text{H}_3\text{PO}_4
ightarrow 3\text{H}_2 + \text{Pb}_3(\text{PO}_4)_2$: A metal replaces hydrogen in an acid, so it is single replacement.
Step4: Identify reaction 4 type
$\text{Li}_3\text{N} + 3\text{NH}_4\text{NO}_3
ightarrow 3\text{LiNO}_3 + (\text{NH}_4)_3\text{N}$: Cations/anions swap between two compounds, so it is double replacement.
Step5: Identify reaction 5 type
$2\text{Na}_2\text{O}
ightarrow 4\text{Na} + \text{O}_2$: One compound breaks into simpler substances, so it is decomposition.
(for mass law question):
Step1: Recall law of conservation of mass
The law states total mass of reactants equals total mass of products.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Reaction Classifications:
- $2\text{NH}_3 + \text{H}_2\text{SO}_4
ightarrow (\text{NH}_4)_2\text{SO}_4$: synthesis
- $4\text{C}_5\text{H}_9\text{O} + 27\text{O}_2
ightarrow 20\text{CO}_2 + 18\text{H}_2\text{O}$: combustion
- $3\text{Pb} + 2\text{H}_3\text{PO}_4
ightarrow 3\text{H}_2 + \text{Pb}_3(\text{PO}_4)_2$: single replacement
- $\text{Li}_3\text{N} + 3\text{NH}_4\text{NO}_3
ightarrow 3\text{LiNO}_3 + (\text{NH}_4)_3\text{N}$: double replacement
- $2\text{Na}_2\text{O}
ightarrow 4\text{Na} + \text{O}_2$: decomposition
Law of Conservation of Mass:
$\text{Mass}_{\text{reactants}} = \text{Mass}_{\text{products}}$ (the last option)