QUESTION IMAGE
Question
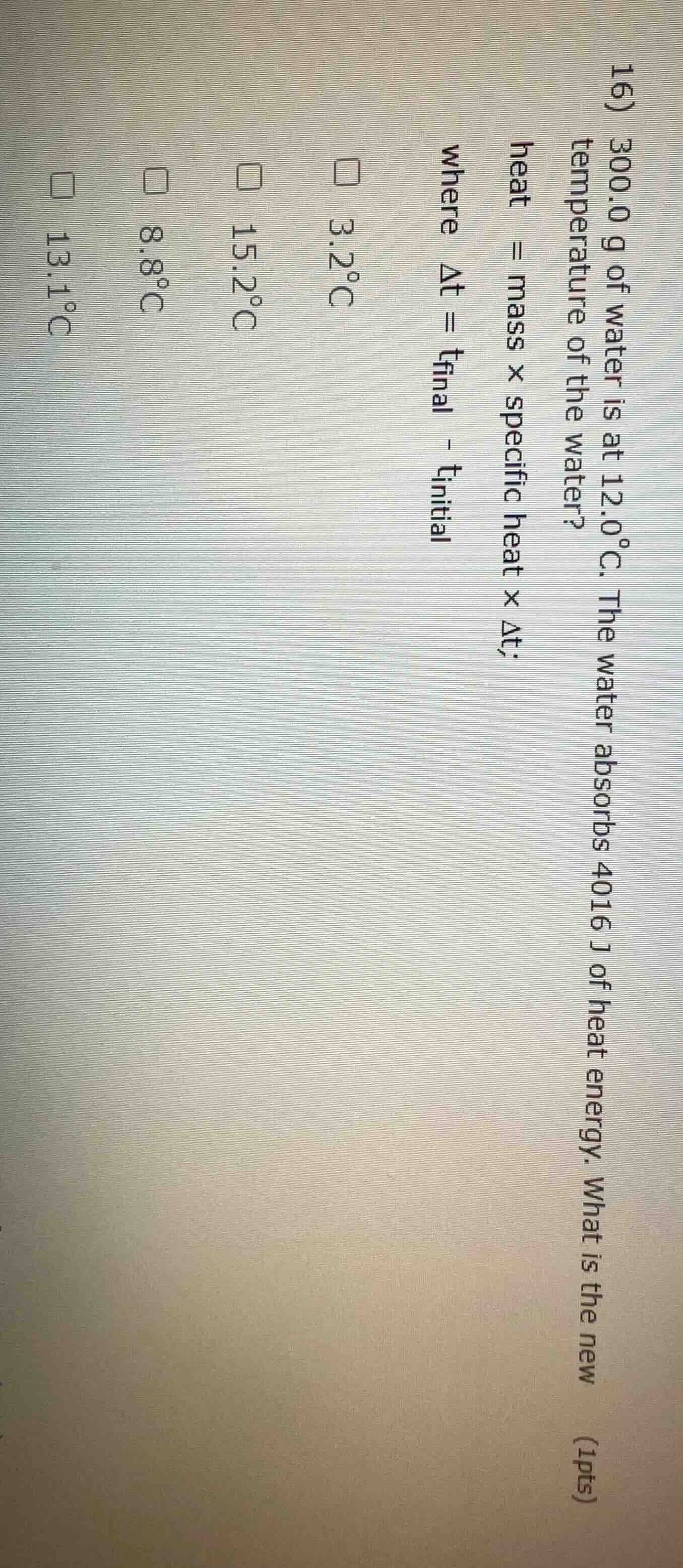
- 300.0 g of water is at 12.0°c. the water absorbs 4016 j of heat energy. what is the new temperature of the water? (1pts) heat = mass × specific heat × δt; where δt = t_final - t_initial
□ 3.2°c
□ 15.2°c
□ 8.8°c
□ 13.1°c
Step1: List known values
$q=4016\ \text{J}$, $m=300.0\ \text{g}$, $c=4.184\ \frac{\text{J}}{\text{g}^\circ\text{C}}$, $t_{\text{initial}}=12.0^\circ\text{C}$
Step2: Rearrange heat formula for $\Delta t$
$\Delta t = \frac{q}{m \times c}$
Step3: Calculate temperature change
$\Delta t = \frac{4016}{300.0 \times 4.184} \approx 3.2^\circ\text{C}$
Step4: Solve for final temperature
$t_{\text{final}} = t_{\text{initial}} + \Delta t$
$t_{\text{final}} = 12.0^\circ\text{C} + 3.2^\circ\text{C} = 15.2^\circ\text{C}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{15.2^\circ\text{C}}$