QUESTION IMAGE
Question
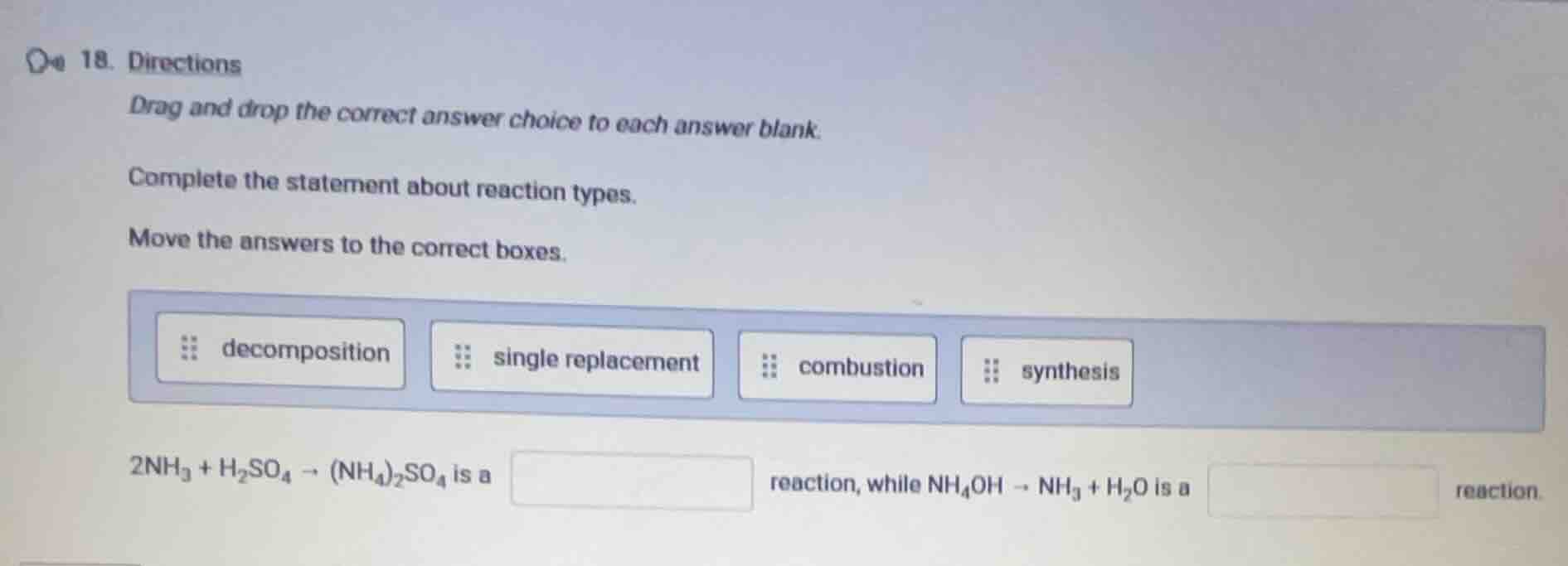
- directions
drag and drop the correct answer choice to each answer blank.
complete the statement about reaction types.
move the answers to the correct boxes.
decomposition single replacement combustion synthesis
$2\ce{nh_3} + \ce{h_2so_4} \
ightarrow (\ce{nh_4})_2\ce{so_4}$ is a \boxed{} reaction, while $\ce{nh_4oh} \
ightarrow \ce{nh_3} + \ce{h_2o}$ is a \boxed{} reaction.
Brief Explanations
- A synthesis reaction combines multiple reactants into one product, which matches the first equation where two compounds form a single salt.
- A decomposition reaction breaks one reactant into multiple products, which matches the second equation where one compound splits into two simpler substances.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $2\text{NH}_3 + \text{H}_2\text{SO}_4
ightarrow (\text{NH}_4)_2\text{SO}_4$ is a synthesis reaction
- $\text{NH}_4\text{OH}
ightarrow \text{NH}_3 + \text{H}_2\text{O}$ is a decomposition reaction