QUESTION IMAGE
Question
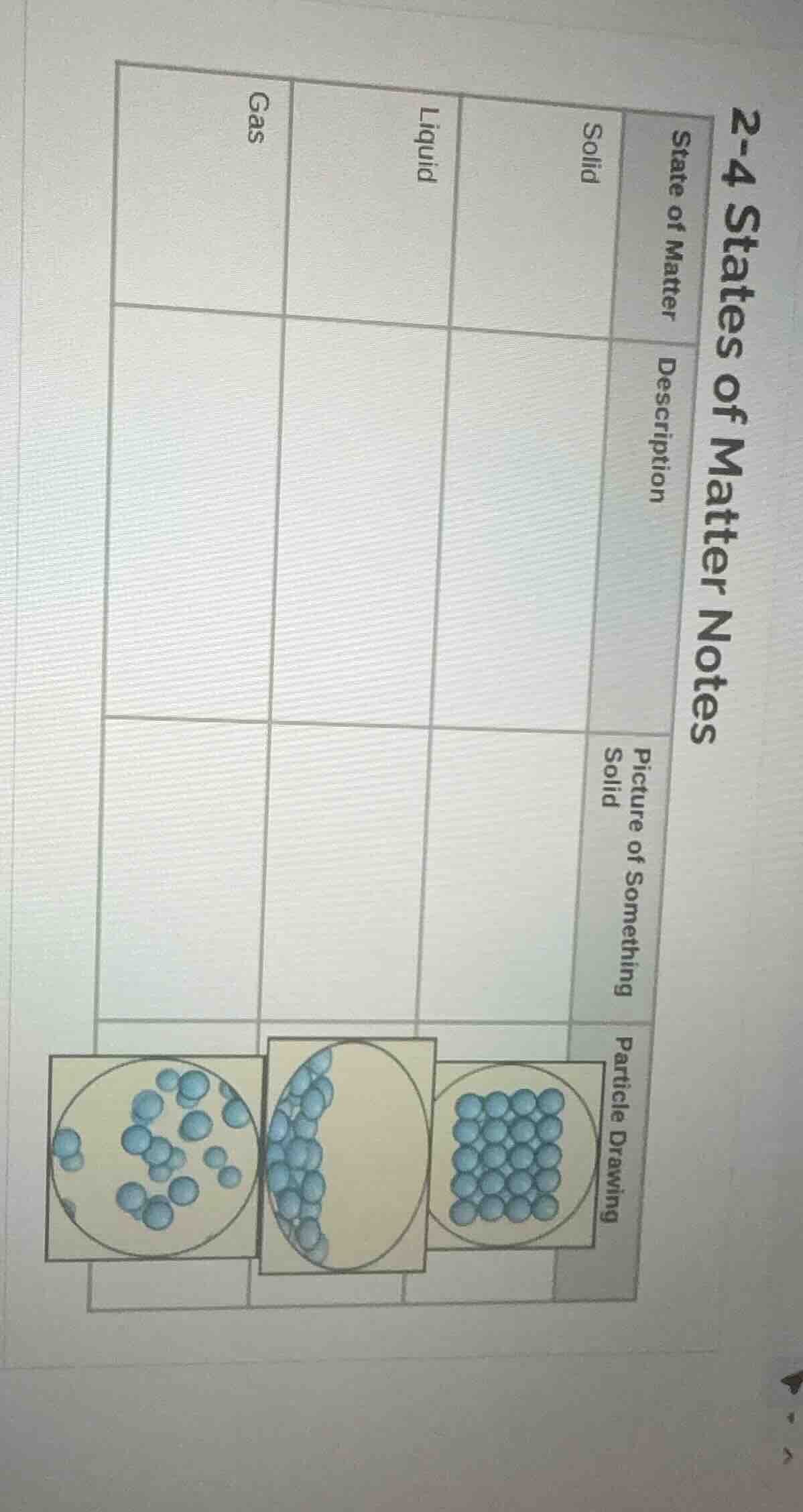
2-4 states of matter notes
state of matter\tdescription\tpicture of something solid\tparticle drawing
\tsolid\t\t
\tliquid\t\t
\tgas\t\t
Brief Explanations
- Solid: Particles are tightly ordered, so solids have rigid, unchanging shapes and set volumes. Common everyday examples include ice or a book.
- Liquid: Particles are close but not locked in place, allowing liquids to flow and match the shape of their container while keeping a consistent volume. Water is the most common example.
- Gas: Particles are spread far apart with lots of space between them, so gases expand to fill any container and have no fixed shape or volume. Air is a mixture of gases. Each state's particle drawing matches these structural properties.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| State of Matter | Description | Picture of Something | Particle Drawing |
|---|---|---|---|
| Liquid | A state of matter with a fixed volume but no fixed shape (takes the shape of its container), where particles are close together but can slide past one another. | Example: Water, juice | <div style="text-align:center;">The middle blue particles clustered at the bottom of the oval, able to flow</div> |
| Gas | A state of matter with no fixed shape or fixed volume (expands to fill its container), where particles are far apart and move freely and quickly. | Example: Air, helium in a balloon | <div style="text-align:center;">The leftmost scattered blue particles spread out across the circle</div> |