QUESTION IMAGE
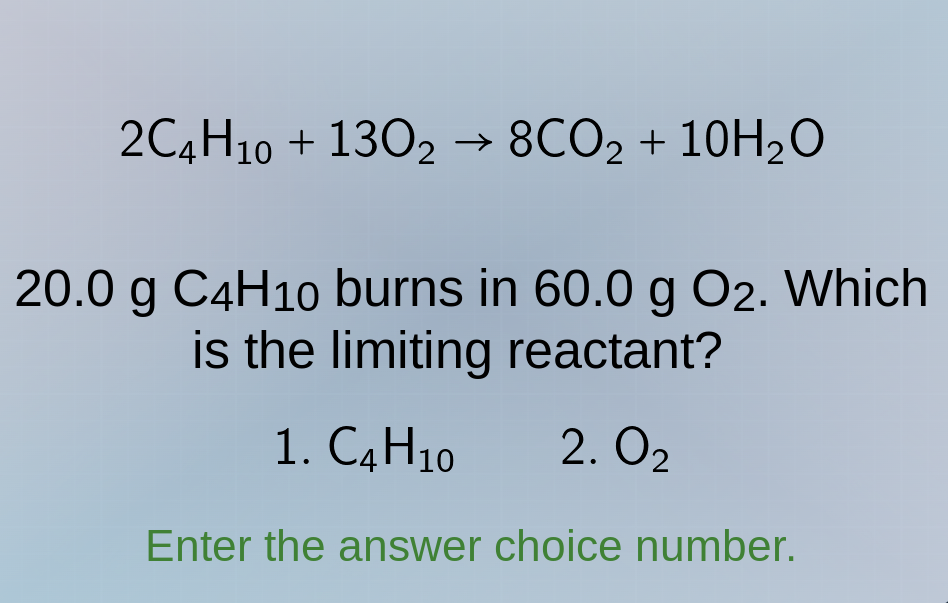
Question
2c₄h₁₀ + 13o₂ → 8co₂ + 10h₂o
20.0 g c₄h₁₀ burns in 60.0 g o₂. which
is the limiting reactant?
- c₄h₁₀ 2. o₂
enter the answer choice number.
Step1: Calculate moles of \( C_4H_{10} \)
Molar mass of \( C_4H_{10} \) is \( 4\times12.01 + 10\times1.008 = 58.12 \, g/mol \). Moles of \( C_4H_{10} = \frac{20.0 \, g}{58.12 \, g/mol} \approx 0.344 \, mol \).
Step2: Calculate moles of \( O_2 \)
Molar mass of \( O_2 \) is \( 32.00 \, g/mol \). Moles of \( O_2 = \frac{60.0 \, g}{32.00 \, g/mol} = 1.875 \, mol \).
Step3: Check mole ratio
From the reaction, \( 2 \, mol \, C_4H_{10} \) reacts with \( 13 \, mol \, O_2 \). For \( 0.344 \, mol \, C_4H_{10} \), required \( O_2 \) is \( \frac{13}{2} \times 0.344 \approx 2.236 \, mol \). But we have only \( 1.875 \, mol \, O_2 \), so \( O_2 \) is limiting? Wait, no, wait. Wait, maybe I reversed. Wait, let's do it the other way. For \( O_2 \), moles available is \( 1.875 \, mol \). The moles of \( C_4H_{10} \) required to react with \( O_2 \) is \( \frac{2}{13} \times 1.875 \approx 0.288 \, mol \). We have \( 0.344 \, mol \, C_4H_{10} \), which is more than \( 0.288 \, mol \). So \( O_2 \) is limiting? Wait, no, wait the options are 1 or 2. Wait, let's recalculate. Wait, molar mass of \( C_4H_{10} \): 412=48, 101=10, total 58. So 20g /58g/mol ≈0.3448 mol. Moles of O2: 60g /32g/mol=1.875 mol. The stoichiometric ratio is 2 C4H10 :13 O2. So moles of O2 needed for 0.3448 mol C4H10: (13/2)0.3448 ≈2.241 mol. But we have only 1.875 mol O2, which is less than 2.241. Wait, no, that would mean O2 is limiting? But wait, another way: moles of C4H10 needed for 1.875 mol O2: (2/13)1.875 ≈0.2885 mol. We have 0.3448 mol C4H10, which is more than 0.2885. So O2 is limiting, so the limiting reactant is O2, which is option 2.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2