QUESTION IMAGE
Question
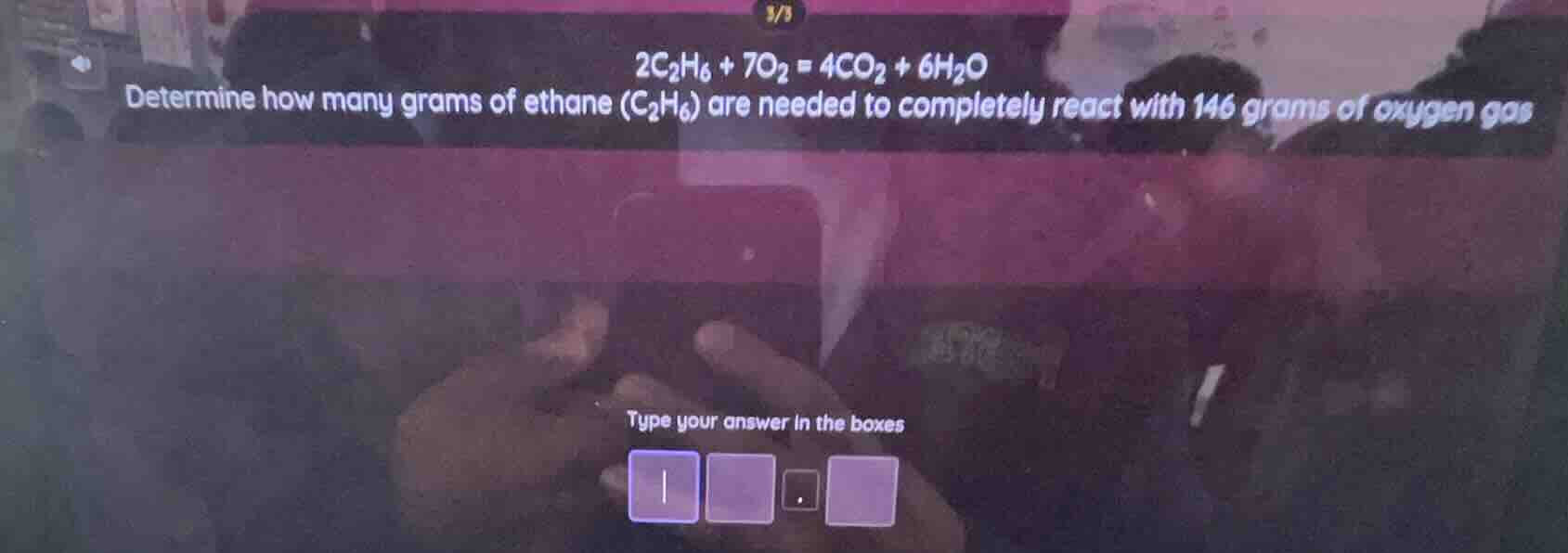
2c₂h₆ + 7o₂ = 4co₂ + 6h₂o determine how many grams of ethane (c₂h₆) are needed to completely react with 146 grams of oxygen gas type your answer in the boxes
Step1: Calculate moles of \(O_2\)
Molar mass of \(O_2\) is \(32\space g/mol\). Moles of \(O_2 = \frac{mass}{molar\space mass}=\frac{146\space g}{32\space g/mol}\approx4.5625\space mol\).
Step2: Relate moles of \(O_2\) to \(C_2H_6\)
From the reaction \(2C_2H_6 + 7O_2 = 4CO_2 + 6H_2O\), the mole ratio of \(C_2H_6\) to \(O_2\) is \(\frac{2}{7}\). Moles of \(C_2H_6=\frac{2}{7}\times moles\space of\space O_2=\frac{2}{7}\times4.5625\space mol\approx1.3036\space mol\).
Step3: Calculate mass of \(C_2H_6\)
Molar mass of \(C_2H_6\) is \(2\times12 + 6\times1 = 30\space g/mol\). Mass of \(C_2H_6 = moles\times molar\space mass = 1.3036\space mol\times30\space g/mol\approx39.11\space g\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(39.1\) (or more precise \(39.11\))