QUESTION IMAGE
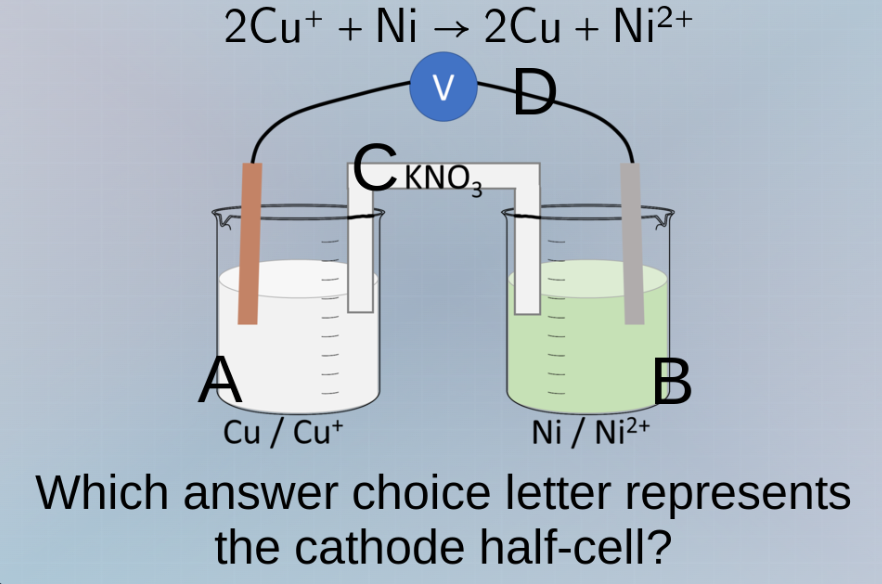
Question
2cu⁺ + ni → 2cu + ni²⁺
which answer choice letter represents the cathode half - cell?
To determine the cathode half - cell, we first analyze the redox reaction. The given reaction is \(2Cu^{+}+Ni
ightarrow2Cu + Ni^{2+}\). In a redox reaction, reduction occurs at the cathode. Reduction is the gain of electrons. Let's look at the oxidation states:
- For \(Cu^{+}\) to \(Cu\), the oxidation state of \(Cu\) changes from + 1 to 0. This means \(Cu^{+}\) is gaining an electron (reduction: \(Cu^{+}+e^{-}
ightarrow Cu\)).
- For \(Ni\) to \(Ni^{2+}\), the oxidation state of \(Ni\) changes from 0 to + 2. This means \(Ni\) is losing electrons (oxidation: \(Ni
ightarrow Ni^{2+}+2e^{-}\)).
In a galvanic cell, the half - cell where reduction occurs is the cathode. The half - cell with \(Cu/Cu^{+}\) (beaker A) is where the reduction of \(Cu^{+}\) to \(Cu\) takes place. The half - cell with \(Ni/Ni^{2+}\) (beaker B) is where oxidation of \(Ni\) to \(Ni^{2+}\) takes place. So the cathode half - cell is the one with \(Cu/Cu^{+}\), which is beaker A.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. \(Cu / Cu^{+}\) (the half - cell in beaker A)