QUESTION IMAGE
Question
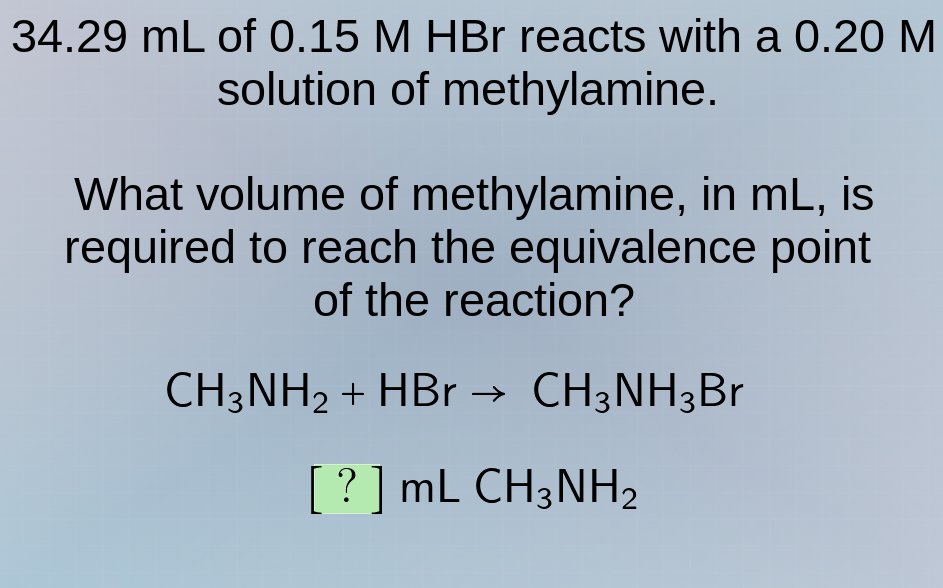
34.29 ml of 0.15 m hbr reacts with a 0.20 m solution of methylamine.
what volume of methylamine, in ml, is required to reach the equivalence point of the reaction?
ch₃nh₂ + hbr → ch₃nh₃br
? ml ch₃nh₂
Step1: Recall the equivalence point concept
At the equivalence point, the moles of acid (HBr) equal the moles of base (methylamine, \(CH_3NH_2\)) according to the balanced chemical equation. The formula for moles is \(n = M\times V\), where \(M\) is molarity and \(V\) is volume in liters.
Step2: Calculate moles of HBr
Given \(V_{HBr}=34.29\space mL = 0.03429\space L\) and \(M_{HBr}=0.15\space M\). Using \(n = M\times V\), we get \(n_{HBr}=0.15\space M\times0.03429\space L = 0.0051435\space mol\).
Step3: Set moles of methylamine equal to moles of HBr
From the balanced equation, the mole ratio of \(CH_3NH_2\) to HBr is \(1:1\). So \(n_{CH_3NH_2}=n_{HBr}=0.0051435\space mol\).
Step4: Calculate volume of methylamine
We know \(M_{CH_3NH_2}=0.20\space M\) and \(n = M\times V\), so \(V=\frac{n}{M}\). Substituting values, \(V_{CH_3NH_2}=\frac{0.0051435\space mol}{0.20\space M}=0.0257175\space L\). Convert to mL: \(0.0257175\space L\times1000\space mL/L = 25.7175\space mL\approx25.72\space mL\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(25.72\)