QUESTION IMAGE
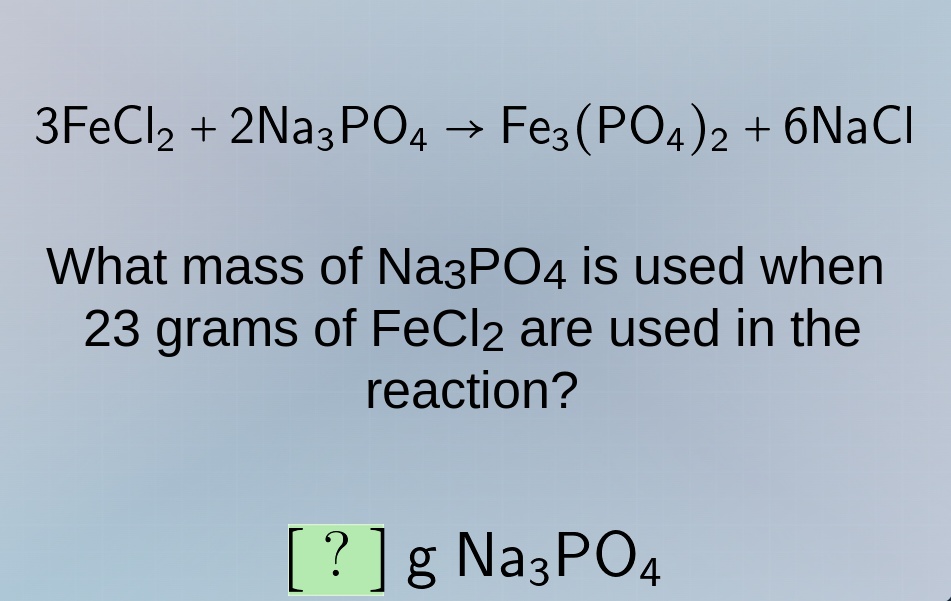
Question
3fecl₂ + 2na₃po₄ → fe₃(po₄)₂ + 6nacl
what mass of na₃po₄ is used when 23 grams of fecl₂ are used in the reaction?
? g na₃po₄
Step1: Calculate moles of \(FeCl_2\)
Molar mass of \(FeCl_2\): \(Fe = 55.85\), \(Cl = 35.45\), so \(M = 55.85 + 2\times35.45 = 126.75\space g/mol\).
Moles of \(FeCl_2 = \frac{23\space g}{126.75\space g/mol} \approx 0.1815\space mol\).
Step2: Use stoichiometry to find moles of \(Na_3PO_4\)
From the reaction: \(3\space mol\space FeCl_2\) reacts with \(2\space mol\space Na_3PO_4\).
Mole ratio: \(\frac{n(Na_3PO_4)}{n(FeCl_2)} = \frac{2}{3}\).
So, \(n(Na_3PO_4) = 0.1815\space mol \times \frac{2}{3} \approx 0.121\space mol\).
Step3: Calculate mass of \(Na_3PO_4\)
Molar mass of \(Na_3PO_4\): \(3\times23 + 31 + 4\times16 = 164\space g/mol\).
Mass = \(0.121\space mol \times 164\space g/mol \approx 19.84\space g\) (or more precisely, re - calculating with more accurate steps:
\(n(FeCl_2)=\frac{23}{126.745}\approx0.1815\)
\(n(Na_3PO_4)=0.1815\times\frac{2}{3}=0.121\)
Mass = \(0.121\times163.94\approx19.84\) (using more accurate molar mass of \(Na_3PO_4 = 163.94\space g/mol\))).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(\approx19.8\) (or more precisely \(\approx19.84\))