QUESTION IMAGE
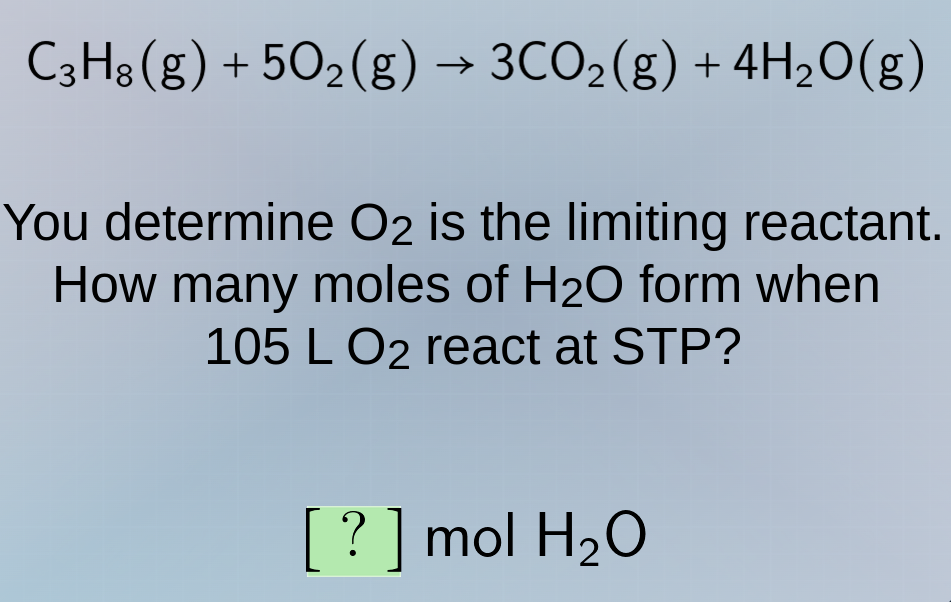
Question
c₃h₈(g) + 5o₂(g) → 3co₂(g) + 4h₂o(g)
you determine o₂ is the limiting reactant.
how many moles of h₂o form when
105 l o₂ react at stp?
? mol h₂o
Step1: Find moles of \( O_2 \) at STP
At STP, 1 mole of any gas occupies 22.4 L. So moles of \( O_2 \) = \( \frac{105\ L}{22.4\ L/mol} \)
Step2: Use stoichiometry from reaction
From the reaction \( \text{C}_3\text{H}_8(\text{g}) + 5\text{O}_2(\text{g})
ightarrow 3\text{CO}_2(\text{g}) + 4\text{H}_2\text{O}(\text{g}) \), 5 moles of \( O_2 \) produce 4 moles of \( H_2O \). Let moles of \( H_2O \) be \( x \). Then \( \frac{x}{\text{moles of } O_2} = \frac{4}{5} \), so \( x = \text{moles of } O_2 \times \frac{4}{5} \)
First, calculate moles of \( O_2 \): \( \frac{105}{22.4} \approx 4.6875\ mol \)
Then, moles of \( H_2O \): \( 4.6875\ mol \times \frac{4}{5} = 3.75\ mol \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
3.75