QUESTION IMAGE
Question
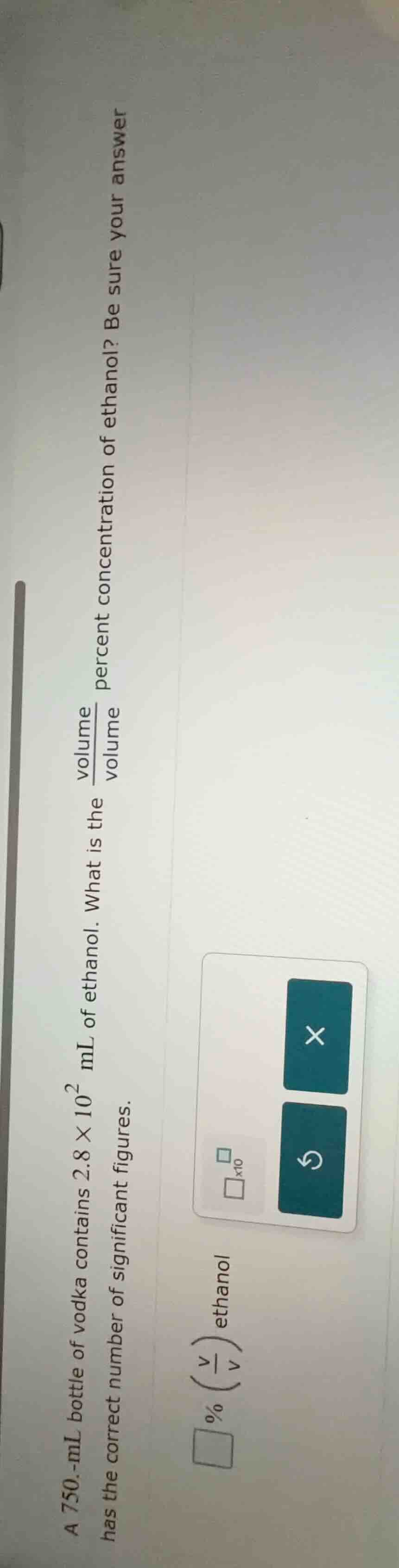
a 750.-ml bottle of vodka contains $2.8 \times 10^{2}$ ml of ethanol. what is the $\frac{volume}{volume}$ percent concentration of ethanol? be sure your answer has the correct number of significant figures. $square$ % $(\frac{v}{v})$ ethanol
Step1: Define v/v% formula
$\text{volume/volume \%} = \frac{\text{volume of solute}}{\text{volume of solution}} \times 100\%$
Step2: Substitute given values
$\text{volume/volume \%} = \frac{2.8 \times 10^2\ \text{mL}}{750.\ \text{mL}} \times 100\%$
Step3: Calculate and round result
$\text{volume/volume \%} = 0.3733... \times 100\% = 37\%$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
37% (v/v) ethanol