QUESTION IMAGE
Question
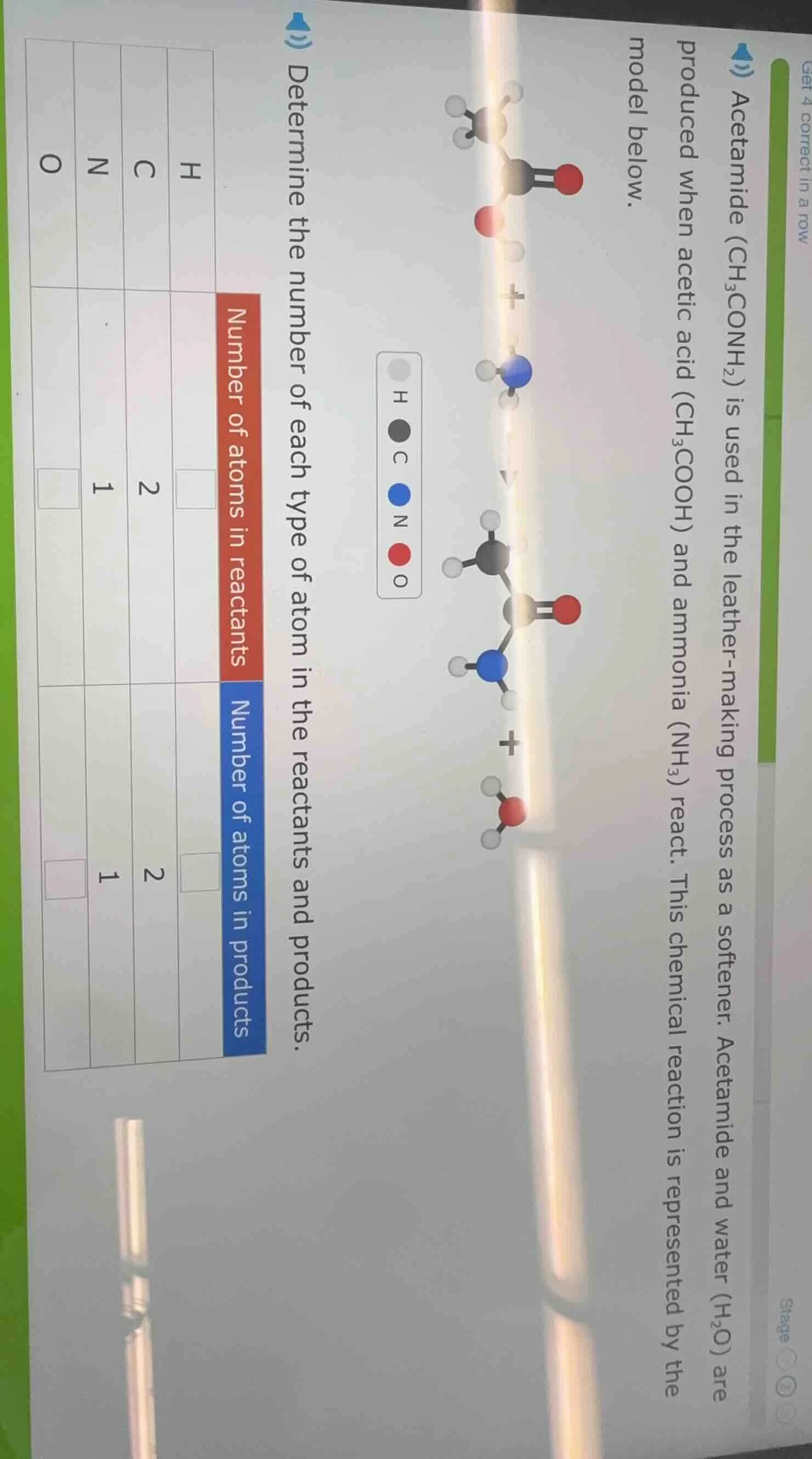
acetamide ($\text{ch}_3\text{conh}_2$) is used in the leather-making process as a softener. acetamide and water ($\text{h}_2\text{o}$) are produced when acetic acid ($\text{ch}_3\text{cooh}$) and ammonia ($\text{nh}_3$) react. this chemical reaction is represented by the model below.determine the number of each type of atom in the reactants and products.| | h | c | n | o || ---- | ---- | ---- | ---- | ---- || number of atoms in reactants | $square$ | 2 | 1 | $square$ || number of atoms in products | $square$ | 2 | 1 | $square$ |(atom key: h=light gray, c=dark gray, n=blue, o=red)
Step1: Count H in reactants
Reactants: $\text{CH}_3\text{COOH}$ has 4 H, $\text{NH}_3$ has 3 H. Total: $4 + 3 = 7$
Step2: Count O in reactants
$\text{CH}_3\text{COOH}$ has 2 O, $\text{NH}_3$ has 0 O. Total: $2 + 0 = 2$
Step3: Count H in products
Products: $\text{CH}_3\text{CONH}_2$ has 5 H, $\text{H}_2\text{O}$ has 2 H. Total: $5 + 2 = 7$
Step4: Count O in products
$\text{CH}_3\text{CONH}_2$ has 1 O, $\text{H}_2\text{O}$ has 1 O. Total: $1 + 1 = 2$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| H | C | N | O | |
|---|---|---|---|---|
| Number of atoms in products | 7 | 2 | 1 | 2 |