QUESTION IMAGE
Question
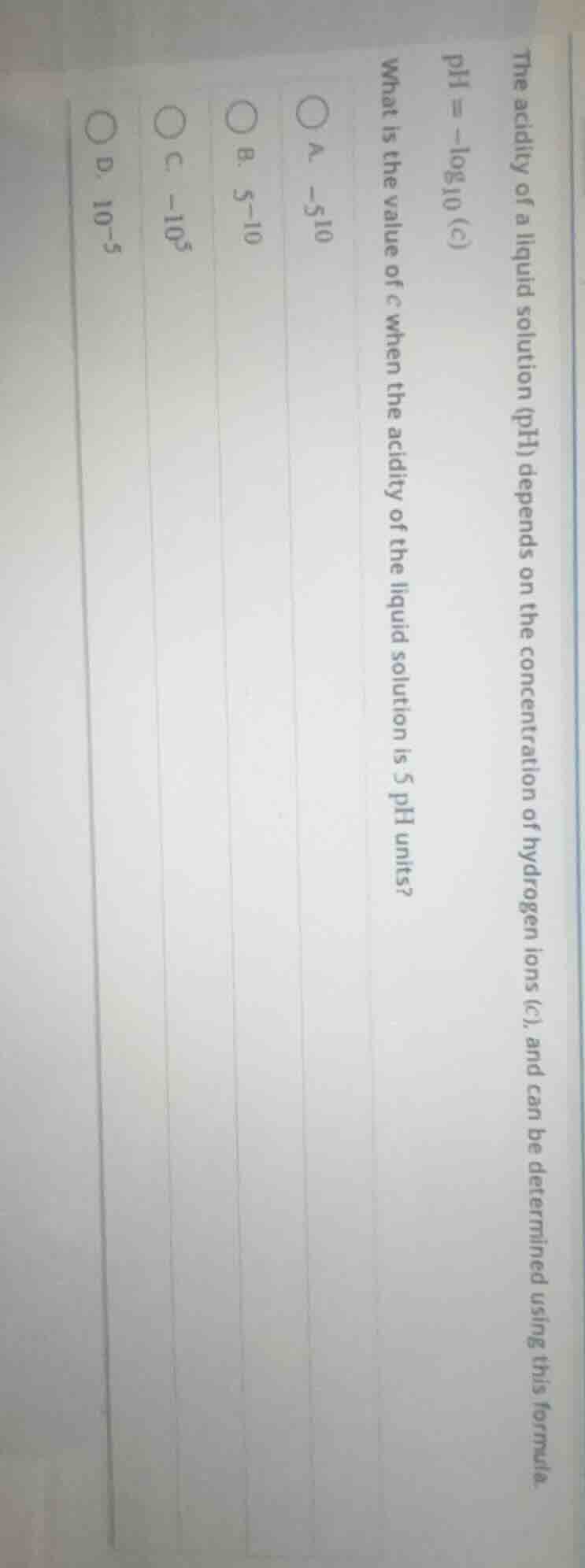
the acidity of a liquid solution (ph) depends on the concentration of hydrogen ions (c), and can be determined using this formula.
$\text{ph} = -\log_{10}(c)$
what is the value of $c$ when the acidity of the liquid solution is 5 ph units?
\\(\circ\\) a. $-5_{10}$
\\(\circ\\) b. $5^{-10}$
\\(\circ\\) c. $-10^5$
\\(\circ\\) d. $10^{-5}$
Step1: Substitute pH=5 into formula
$5 = -\log_{10}(c)$
Step2: Rearrange to isolate log term
$\log_{10}(c) = -5$
Step3: Convert to exponential form
$c = 10^{-5}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. $10^{-5}$